This regulatory network was inferred from the input dataset. The miRNAs and mRNAs are
presented as round and rectangle nodes respectively. The numerical value popped up upon mouse over the gene node is the log2 transformed fold-change of the gene expression between the two groups. All of the nodes are clickable, and the detailed information of the miRNAs/mRNAs and related cancer pathway will be displayed in another window. The edges between nodes are supported by both interactions (predicted or experimentally verified) and correlations learnt from cancer dataset. The numerical value popped up upon mouse over the edge is the correlation beat value (effect size) between the two nodes. The experimental evidences of the edges reported in previous cancer studies are highlighted by red/orange color. All of these information can be accessed by the "mouse-over" action. This network shows a full map of the miRNA-mRNA regulation of the input gene list(s), and the hub miRNAs (with the high network degree/betweenness centrality) would be the potential cancer drivers or tumor suppressors. The full result table can be accessed in the "Regulations" tab.
"miRNACancerMAP" is also a network visualization tool for users to draw their regulatory network by personal customization. Users can set the complexity of the network by limiting the number of nodes or edges. And the color of the nodes can be defined by different categories of the mRNAs and miRNAs, such as Gene-Ontology, pathway, and expression status. Users can also select to use network degree or network betweenness centrality to define the node size. And edges can be black or colored by the correlation. Purple edge means negative correlation (mostly found between miRNA and mRNA), and blue edge means positive correlation (found in PPI or miRNA-miRNA sponge effect). We can also add the protein-protein interactions (PPI) into the network. This result will show the cluster of genes regulated by some specific miRNAs. Additionally, miRNA-miRNA edges can be added by the "miRNA sponge" button, presenting some clusters of miRNAs that have the interactions via sponge effect.
miRNA-gene regulations
| Num | microRNA | Gene | miRNA log2FC | miRNA pvalue | Gene log2FC | Gene pvalue | Interaction | Correlation beta | Correlation P-value | PMID | Reported in cancer studies |
|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | hsa-miR-23a-3p | APAF1 | 1.11 | 0 | 0.3 | 0.22769 | miRNATAP | -0.49 | 0 | 24992592; 24249161 | Luciferase assay was performed to verify a putative target site of miR-23a in the 3'-UTR of apoptosis protease activating factor 1 APAF1 mRNA; The expression levels of miR-23a and APAF1 in CRC cell lines SW480 and SW620 and clinical samples were assessed using reverse transcription-quantitative real-time PCR RT-qPCR and Western blot; Moreover miR-23a up-regulation was coupled with APAF1 down-regulation in CRC tissue samples;We found that the expression of miR-23a was increased and the level of apoptosis-activating factor-1 APAF-1 was decreased in 5-FU-treated colon cancer cells compared to untreated cells; APAF-1 as a target gene of miR-23a was identified and miR-23a antisense-induced increase in the activation of caspase-9 was observed |
| 2 | hsa-miR-23b-3p | APAF1 | -0.29 | 0.18665 | 0.3 | 0.22769 | miRNATAP | -0.54 | 0 | NA | |
| 3 | hsa-miR-27a-3p | APAF1 | 1.3 | 0 | 0.3 | 0.22769 | miRNATAP | -0.39 | 0 | NA | |
| 4 | hsa-miR-27b-3p | APAF1 | 0.08 | 0.72527 | 0.3 | 0.22769 | miRNATAP | -0.48 | 0 | NA | |
| 5 | hsa-miR-708-3p | APAF1 | 0.62 | 0.27536 | 0.3 | 0.22769 | mirMAP | -0.24 | 0 | NA | |
| 6 | hsa-miR-944 | APAF1 | 3.33 | 0.01778 | 0.3 | 0.22769 | miRNATAP | -0.13 | 0 | NA | |
| 7 | hsa-miR-125a-3p | ATM | 1.2 | 0.00164 | -0.39 | 0.08341 | miRanda | -0.13 | 0.00188 | NA | |
| 8 | hsa-miR-203a-3p | ATM | 1.45 | 0.03941 | -0.39 | 0.08341 | MirTarget | -0.14 | 0 | 24145123; 27542403 | miR 203 induces oxaliplatin resistance in colorectal cancer cells by negatively regulating ATM kinase; In silico analysis identified ataxia telangiectasia mutated ATM a primary mediator of the DNA damage response as a potential target of miR-203; Using TCGA database we identified a significant reverse correlation of miR-203 and ATM expression in CRC tissues; We validated ATM as a bona fide target of miR-203 in CRC cells; Mutation of the putative miR-203 binding site in the 3' untranslated region 3'UTR of the ATM mRNA abolished the inhibitory effect of miR-203 on ATM;MiR 203 inhibits tumor invasion and metastasis in gastric cancer by ATM; Our results showed that miR-203 was significantly downregulated in gastric cancer tissues and cells while ataxia telangiectasia mutated kinase ATM was upregulated in gastric cancer tissues and cells and was directly regulated by miR-203; ATM knockdown phenocopied the effect of miR-203 overexpression |
| 9 | hsa-miR-590-5p | ATM | 1.04 | 0.00027 | -0.39 | 0.08341 | mirMAP | -0.17 | 0.00247 | NA | |
| 10 | hsa-miR-30b-5p | ATR | -0.43 | 0.05936 | 0.73 | 0.00017 | mirMAP | -0.19 | 0.00265 | NA | |
| 11 | hsa-miR-30a-5p | BAX | -1.72 | 0 | 1.23 | 0 | miRNAWalker2 validate | -0.15 | 0.00018 | NA | |
| 12 | hsa-miR-365a-3p | BAX | -0.89 | 0.00255 | 1.23 | 0 | miRNAWalker2 validate | -0.19 | 6.0E-5 | 24216611 | MiR 365 induces gemcitabine resistance in pancreatic cancer cells by targeting the adaptor protein SHC1 and pro apoptotic regulator BAX |
| 13 | hsa-let-7a-2-3p | BBC3 | 0.2 | 0.64356 | 1.16 | 0.00014 | MirTarget | -0.19 | 0.00032 | NA | |
| 14 | hsa-miR-149-5p | BBC3 | 0.71 | 0.29685 | 1.16 | 0.00014 | miRNATAP | -0.13 | 7.0E-5 | NA | |
| 15 | hsa-miR-27b-3p | BBC3 | 0.08 | 0.72527 | 1.16 | 0.00014 | miRNATAP | -0.25 | 0.00693 | NA | |
| 16 | hsa-miR-129-5p | CASP8 | -2.57 | 0 | 1.08 | 0 | miRanda | -0.14 | 0 | NA | |
| 17 | hsa-miR-143-3p | CASP8 | -0.66 | 0.04832 | 1.08 | 0 | MirTarget | -0.19 | 3.0E-5 | NA | |
| 18 | hsa-miR-193b-3p | CASP9 | 0.28 | 0.45126 | -0.31 | 0.11004 | miRNAWalker2 validate | -0.17 | 0 | NA | |
| 19 | hsa-miR-139-5p | CCNB1 | -1.83 | 0 | 2.86 | 0 | miRanda | -0.49 | 0 | NA | |
| 20 | hsa-miR-338-3p | CCND1 | -0.96 | 0.01915 | 0.62 | 0.1312 | miRNAWalker2 validate; miRTarBase; miRanda | -0.24 | 0.00086 | NA | |
| 21 | hsa-let-7a-3p | CCND2 | 0.5 | 0.04111 | -0.5 | 0.3 | mirMAP | -0.54 | 0.0001 | 20418948 | MicroRNA let 7a inhibits proliferation of human prostate cancer cells in vitro and in vivo by targeting E2F2 and CCND2 |
| 22 | hsa-let-7b-3p | CCND2 | 0.22 | 0.29604 | -0.5 | 0.3 | mirMAP | -0.46 | 0.00588 | NA | |
| 23 | hsa-miR-106b-5p | CCND2 | 1.71 | 0 | -0.5 | 0.3 | miRNAWalker2 validate; miRTarBase; miRNATAP | -0.42 | 9.0E-5 | NA | |
| 24 | hsa-miR-130b-5p | CCND2 | 0.7 | 0.05101 | -0.5 | 0.3 | mirMAP | -0.39 | 2.0E-5 | NA | |
| 25 | hsa-miR-141-3p | CCND2 | 1.46 | 0.00116 | -0.5 | 0.3 | MirTarget; TargetScan | -0.21 | 0.00436 | NA | |
| 26 | hsa-miR-15b-5p | CCND2 | 1.62 | 0 | -0.5 | 0.3 | miRNATAP | -0.44 | 0.00021 | NA | |
| 27 | hsa-miR-16-2-3p | CCND2 | 1.8 | 0 | -0.5 | 0.3 | mirMAP | -0.55 | 0 | NA | |
| 28 | hsa-miR-16-5p | CCND2 | 1.01 | 1.0E-5 | -0.5 | 0.3 | miRNAWalker2 validate; miRNATAP | -0.67 | 1.0E-5 | NA | |
| 29 | hsa-miR-181a-2-3p | CCND2 | 0.9 | 0.00083 | -0.5 | 0.3 | mirMAP | -0.38 | 0.00236 | NA | |
| 30 | hsa-miR-182-5p | CCND2 | 0.89 | 0.03106 | -0.5 | 0.3 | miRNAWalker2 validate; miRTarBase; miRNATAP | -0.32 | 9.0E-5 | NA | |
| 31 | hsa-miR-183-5p | CCND2 | 1.66 | 0.00052 | -0.5 | 0.3 | miRNATAP | -0.29 | 2.0E-5 | NA | |
| 32 | hsa-miR-19b-3p | CCND2 | 0.76 | 0.00653 | -0.5 | 0.3 | miRNAWalker2 validate; MirTarget; miRNATAP | -0.32 | 0.00853 | NA | |
| 33 | hsa-miR-21-3p | CCND2 | 1.75 | 0 | -0.5 | 0.3 | mirMAP | -0.33 | 0.0051 | NA | |
| 34 | hsa-miR-224-3p | CCND2 | 1.52 | 0.0065 | -0.5 | 0.3 | mirMAP | -0.27 | 4.0E-5 | NA | |
| 35 | hsa-miR-26b-5p | CCND2 | -0.3 | 0.16008 | -0.5 | 0.3 | mirMAP; miRNATAP | -0.58 | 0.00034 | NA | |
| 36 | hsa-miR-301a-3p | CCND2 | 1.45 | 1.0E-5 | -0.5 | 0.3 | miRNAWalker2 validate | -0.38 | 0.0002 | NA | |
| 37 | hsa-miR-3065-3p | CCND2 | -1.04 | 0.02184 | -0.5 | 0.3 | MirTarget; miRNATAP | -0.2 | 0.00685 | NA | |
| 38 | hsa-miR-3065-5p | CCND2 | -0.24 | 0.63312 | -0.5 | 0.3 | mirMAP | -0.24 | 0.00057 | NA | |
| 39 | hsa-miR-424-5p | CCND2 | 1.09 | 0.00042 | -0.5 | 0.3 | miRNATAP | -0.39 | 0.0003 | NA | |
| 40 | hsa-miR-429 | CCND2 | 1.4 | 0.009 | -0.5 | 0.3 | miRNATAP | -0.19 | 0.002 | NA | |
| 41 | hsa-miR-450b-5p | CCND2 | 0.46 | 0.13274 | -0.5 | 0.3 | MirTarget; PITA; miRNATAP | -0.32 | 0.00413 | NA | |
| 42 | hsa-miR-590-3p | CCND2 | 1.12 | 0.00016 | -0.5 | 0.3 | miRanda; mirMAP | -0.33 | 0.00348 | NA | |
| 43 | hsa-miR-590-5p | CCND2 | 1.04 | 0.00027 | -0.5 | 0.3 | mirMAP | -0.41 | 0.00055 | NA | |
| 44 | hsa-miR-9-3p | CCND2 | 0.33 | 0.54111 | -0.5 | 0.3 | MirTarget; mirMAP; miRNATAP | -0.19 | 0.00361 | NA | |
| 45 | hsa-miR-93-5p | CCND2 | 1.75 | 0 | -0.5 | 0.3 | miRNATAP | -0.32 | 0.00244 | NA | |
| 46 | hsa-miR-96-5p | CCND2 | 1.14 | 0.00943 | -0.5 | 0.3 | TargetScan; miRNATAP | -0.33 | 3.0E-5 | NA | |
| 47 | hsa-miR-96-5p | CCND3 | 1.14 | 0.00943 | 0.04 | 0.88352 | TargetScan | -0.12 | 0.00328 | NA | |
| 48 | hsa-miR-125b-5p | CCNE1 | -0.51 | 0.13327 | 2.19 | 0 | miRNAWalker2 validate | -0.26 | 0.00155 | NA | |
| 49 | hsa-miR-195-5p | CCNE1 | -0.91 | 0.00151 | 2.19 | 0 | miRNAWalker2 validate; MirTarget; miRNATAP | -0.43 | 1.0E-5 | 24402230 | Furthermore through qPCR and western blot assays we showed that overexpression of miR-195-5p reduced CCNE1 mRNA and protein levels respectively |
| 50 | hsa-miR-497-5p | CCNE1 | -0.8 | 0.0036 | 2.19 | 0 | MirTarget; miRNATAP | -0.43 | 2.0E-5 | 24112607; 25909221; 24909281 | Western blot assays confirmed that overexpression of miR-497 reduced cyclin E1 protein levels; Inhibited cellular growth suppressed cellular migration and invasion and G1 cell cycle arrest were observed upon overexpression of miR-497 in cells possibly by targeting cyclin E1;The effect of simultaneous overexpression of miR-497 and miR-34a on the inhibition of cell proliferation colony formation and tumor growth and the downregulation of cyclin E1 was stronger than the effect of each miRNA alone; The synergistic actions of miR-497 and miR-34a partly correlated with cyclin E1 levels; These results indicate cyclin E1 is downregulated by both miR-497 and miR-34a which synergistically retard the growth of human lung cancer cells;miR 497 suppresses proliferation of human cervical carcinoma HeLa cells by targeting cyclin E1; Furthermore the target effect of miR-497 on the CCNE1 was identified by dual-luciferase reporter assay system qRT-PCR and Western blotting; Over-expressed miR-497 in HeLa cells could suppress cell proliferation by targeting CCNE1 |
| 51 | hsa-miR-26a-5p | CCNE2 | -0.38 | 0.04425 | 1.36 | 0 | miRNAWalker2 validate; miRTarBase; miRNATAP | -0.44 | 0.0001 | 24116110; 21901171 | The loss of miR 26a mediated post transcriptional regulation of cyclin E2 in pancreatic cancer cell proliferation and decreased patient survival; The in vitro and in vivo assays showed that overexpression of miR-26a resulted in cell cycle arrest inhibited cell proliferation and decreased tumor growth which was associated with cyclin E2 downregulation;We also show that enforced expression of miR-26a in AML cells is able to inhibit cell cycle progression by downregulating cyclin E2 expression |
| 52 | hsa-miR-30a-5p | CCNE2 | -1.72 | 0 | 1.36 | 0 | miRNATAP | -0.22 | 0.00034 | NA | |
| 53 | hsa-let-7b-5p | CCNG1 | 0.06 | 0.7814 | -1.13 | 0 | miRNAWalker2 validate | -0.28 | 0.00054 | NA | |
| 54 | hsa-miR-1271-5p | CCNG1 | 1.16 | 0.00208 | -1.13 | 0 | MirTarget | -0.18 | 6.0E-5 | 26477861 | MiR 1271 Inhibits Ovarian Cancer Growth by Targeting Cyclin G1; MiR-1271 exerted its role by targeting CCNG1 |
| 55 | hsa-miR-21-5p | CCNG1 | 1.75 | 0 | -1.13 | 0 | miRNAWalker2 validate | -0.35 | 6.0E-5 | NA | |
| 56 | hsa-miR-23a-3p | CCNG1 | 1.11 | 0 | -1.13 | 0 | MirTarget; miRNATAP | -0.53 | 0 | NA | |
| 57 | hsa-miR-23b-3p | CCNG1 | -0.29 | 0.18665 | -1.13 | 0 | MirTarget; miRNATAP | -0.23 | 0.00341 | 26872615 | MiR 23b targets cyclin G1 and suppresses ovarian cancer tumorigenesis and progression; Dual-luciferase reporter assay and a xenograft mouse model were used to examine the expression of miR-23b and its target gene CCNG1; Dual-luciferase reporter assay showed that miR-23b bound with the 3' untranslated region of CCNG1; Furthermore miR-23b inhibited tumor growth and suppressed CCNG1 expression in vitro; Our findings show that miR-23b may inhibit ovarian cancer tumorigenesis and progression by downregulating CCNG1 and the expression of the relevant genes |
| 58 | hsa-miR-24-3p | CCNG1 | 1.09 | 0 | -1.13 | 0 | miRNAWalker2 validate | -0.46 | 0 | NA | |
| 59 | hsa-miR-27a-3p | CCNG1 | 1.3 | 0 | -1.13 | 0 | MirTarget; miRNATAP | -0.44 | 0 | NA | |
| 60 | hsa-miR-27b-3p | CCNG1 | 0.08 | 0.72527 | -1.13 | 0 | MirTarget; miRNATAP | -0.28 | 6.0E-5 | 26623719 | Moreover miR-27b directly targets the 3' untranslated regions 3'-UTRs of CCNG1 a well-known negative regulator of P53 stability; Interestingly miR-27b up-regulation leads to increased miR-508-5p expression and this phenomenon is mediated by CCNG1 and P53 |
| 61 | hsa-miR-299-5p | CCNG1 | -0.17 | 0.63894 | -1.13 | 0 | MirTarget | -0.18 | 7.0E-5 | NA | |
| 62 | hsa-miR-409-3p | CCNG1 | 0.65 | 0.05151 | -1.13 | 0 | MirTarget; PITA; miRNATAP | -0.16 | 0.0017 | NA | |
| 63 | hsa-let-7f-1-3p | CCNG2 | 1.29 | 0 | -0.2 | 0.44774 | MirTarget; mirMAP | -0.28 | 6.0E-5 | NA | |
| 64 | hsa-miR-503-5p | CCNG2 | 1.89 | 0 | -0.2 | 0.44774 | miRNAWalker2 validate | -0.17 | 0.0003 | NA | |
| 65 | hsa-miR-145-5p | CDK4 | -1.75 | 2.0E-5 | 1.16 | 0 | miRNAWalker2 validate; miRTarBase | -0.13 | 0.00065 | 21092188 | Furthermore we found that CDK4 was regulated by miR-145 in cell cycle control |
| 66 | hsa-miR-195-5p | CDK4 | -0.91 | 0.00151 | 1.16 | 0 | miRNAWalker2 validate; miRTarBase | -0.16 | 0.00344 | NA | |
| 67 | hsa-miR-101-3p | CDK6 | -0.45 | 0.02834 | 0.3 | 0.4008 | mirMAP | -0.39 | 0.00173 | NA | |
| 68 | hsa-miR-129-5p | CDK6 | -2.57 | 0 | 0.3 | 0.4008 | miRNAWalker2 validate | -0.13 | 0.00427 | 24055727 | Interestingly we showed that cyclin dependent kinase 6 CDK6 a cell cycle-associated protein involved in G1-S transition was a target of miR-129 |
| 69 | hsa-miR-30d-3p | CDK6 | -0.55 | 0.04337 | 0.3 | 0.4008 | mirMAP | -0.28 | 0.00298 | NA | |
| 70 | hsa-miR-30d-5p | CDK6 | -0.55 | 0.01401 | 0.3 | 0.4008 | mirMAP | -0.44 | 9.0E-5 | NA | |
| 71 | hsa-let-7d-5p | CDKN1A | 0.98 | 0 | -0.53 | 0.09948 | MirTarget | -0.4 | 0.00048 | NA | |
| 72 | hsa-let-7g-5p | CDKN1A | 0.33 | 0.24114 | -0.53 | 0.09948 | MirTarget | -0.31 | 0.00013 | NA | |
| 73 | hsa-let-7i-5p | CDKN1A | 0.8 | 3.0E-5 | -0.53 | 0.09948 | MirTarget | -0.32 | 0.00701 | NA | |
| 74 | hsa-miR-101-3p | CDKN1A | -0.45 | 0.02834 | -0.53 | 0.09948 | MirTarget | -0.45 | 6.0E-5 | NA | |
| 75 | hsa-miR-106b-5p | CDKN1A | 1.71 | 0 | -0.53 | 0.09948 | miRNAWalker2 validate; miRTarBase; MirTarget; miRNATAP | -0.32 | 1.0E-5 | NA | |
| 76 | hsa-miR-146a-5p | CDKN1A | 1.82 | 2.0E-5 | -0.53 | 0.09948 | miRNAWalker2 validate | -0.18 | 0.00045 | NA | |
| 77 | hsa-miR-17-5p | CDKN1A | 1.66 | 0 | -0.53 | 0.09948 | miRNAWalker2 validate; miRTarBase; MirTarget; TargetScan; miRNATAP | -0.26 | 0.00022 | 26482648; 24989082 | The low expressions of miR-17 and miR-92 families can maintain cisplatin resistance through the regulation of CDKN1A and RAD21;According to PicTar and Miranda algorithms which predicted CDKN1A p21 as a putative target of miR-17 a luciferase assay was performed and revealed that miR-17 directly targets the 3'-UTR of p21 mRNA |
| 78 | hsa-miR-20a-5p | CDKN1A | 1.45 | 0 | -0.53 | 0.09948 | miRNAWalker2 validate; miRTarBase; MirTarget; miRNATAP | -0.31 | 4.0E-5 | 26012475 | Using the poorly tumorigenic and TGF-β-sensitive FET cell line that expresses low miR-20a levels we first confirmed that miR-20a downmodulated CDKN1A expression both at mRNA and protein level through direct binding to its 3'-UTR; Moreover besides modulating CDKN1A miR-20a blocked TGF-β-induced transactivation of its promoter without affecting the post-receptor activation of Smad3/4 effectors directly; Finally miR-20a abrogated the TGF-β-mediated c-Myc repression a direct inhibitor of the CDKN1A promoter activation most likely by reducing the expression of specific MYC-regulating genes from the Smad/E2F-based core repressor complex |
| 79 | hsa-miR-30b-3p | CDKN1A | -0.27 | 0.40085 | -0.53 | 0.09948 | MirTarget | -0.23 | 0.00125 | NA | |
| 80 | hsa-miR-335-5p | CDKN1A | 1.6 | 6.0E-5 | -0.53 | 0.09948 | miRNAWalker2 validate | -0.32 | 0 | NA | |
| 81 | hsa-miR-345-5p | CDKN1A | 1.84 | 0 | -0.53 | 0.09948 | miRNAWalker2 validate; miRTarBase; MirTarget | -0.24 | 0.00033 | NA | |
| 82 | hsa-miR-423-3p | CDKN1A | 0.82 | 0.00026 | -0.53 | 0.09948 | miRNAWalker2 validate; miRTarBase | -0.32 | 0.00138 | NA | |
| 83 | hsa-miR-423-5p | CDKN1A | 0.83 | 0.00079 | -0.53 | 0.09948 | MirTarget | -0.41 | 0 | NA | |
| 84 | hsa-miR-429 | CDKN1A | 1.4 | 0.009 | -0.53 | 0.09948 | miRNATAP | -0.13 | 0.00128 | NA | |
| 85 | hsa-miR-93-5p | CDKN1A | 1.75 | 0 | -0.53 | 0.09948 | miRNAWalker2 validate; miRTarBase; MirTarget; miRNATAP | -0.32 | 1.0E-5 | 25633810 | MicroRNA 93 activates c Met/PI3K/Akt pathway activity in hepatocellular carcinoma by directly inhibiting PTEN and CDKN1A; We confirmed that miR-93 directly bound with the 3' untranslated regions of the tumor-suppressor genes PTEN and CDKN1A respectivelyand inhibited their expression; We concluded that miR-93 stimulated cell proliferation migration and invasion through the oncogenic c-Met/PI3K/Akt pathway and also inhibited apoptosis by directly inhibiting PTEN and CDKN1A expression in human HCC |
| 86 | hsa-miR-942-5p | CDKN1A | 1.91 | 0 | -0.53 | 0.09948 | miRNAWalker2 validate | -0.18 | 0.00704 | NA | |
| 87 | hsa-miR-125b-5p | CDKN2A | -0.51 | 0.13327 | 1.06 | 0.26133 | miRNAWalker2 validate | -0.58 | 0.00347 | 23585871 | In this study we further extend our studies by showing that miR-125b represses the protein product of the ink4a/ARF locus p14ARF in two prostate cancer cell lines LNCaP wild type-p53 and 22Rv1 both wild type and mutant p53 as well as in the PC-346C prostate cancer xenograft model that lentivirally overexpressed miR-125b; Conversely treatment of prostate cancer cells with an inhibitor of miR-125b anti-miR-125b resulted in increased expression of p14ARF decreased level of Mdm2 and induction of apoptosis; In addition overexpression of miR-125b in p53-deficient PC3 cells induced down-regulation of p14ARF which leads to increased cell proliferation through a p53-independent manner |
| 88 | hsa-miR-365a-3p | CDKN2A | -0.89 | 0.00255 | 1.06 | 0.26133 | MirTarget | -0.76 | 0.00074 | NA | |
| 89 | hsa-miR-129-5p | CHEK1 | -2.57 | 0 | 2.16 | 0 | miRanda | -0.14 | 0.00016 | NA | |
| 90 | hsa-miR-139-5p | CHEK1 | -1.83 | 0 | 2.16 | 0 | miRanda | -0.39 | 0 | NA | |
| 91 | hsa-miR-195-5p | CHEK1 | -0.91 | 0.00151 | 2.16 | 0 | MirTarget; miRNATAP | -0.34 | 0 | 25840419 | MiR 195 suppresses non small cell lung cancer by targeting CHEK1; We discovered that CHEK1 was a direct target of miR-195 which decreased CHEK1 expression in lung cancer cells |
| 92 | hsa-miR-497-5p | CHEK1 | -0.8 | 0.0036 | 2.16 | 0 | MirTarget; miRNATAP | -0.35 | 0 | 24464213 | Checkpoint kinase 1 is negatively regulated by miR 497 in hepatocellular carcinoma; In silico analysis showed that CHEK1 was a candidate target of miR-497 which was previously found to be downregulated in HCC by us; To test whether miR-497 could bind to 3'untranslated region 3'UTR of CHEK1 luciferase reporter assay was conducted; The result revealed that miR-497 could bind to the 3'untranslated region 3'UTR of CHEK1 mRNA; Western blot showed that ectopic expression of miR-497 suppressed the CHEK1 expression and inhibition of miR-497 led to significant upregulation of CHEK1; Finally miR-497 expression was measured in the same 30 HCC samples and the correlation between miR-497 and CHEK1 was analyzed; The results indicated that miR-497 was downregulated in HCC and had a significant negative correlation with CHEK1; Taken together these results demonstrated that CHEK1 was negatively regulated by miR-497 and the overexpressed CHEK1 was resulted from the downregulated miR-497 in HCC which provided a potential molecular target for HCC therapy |
| 93 | hsa-miR-342-3p | CYCS | 0.32 | 0.26915 | 0.2 | 0.35913 | mirMAP | -0.21 | 8.0E-5 | NA | |
| 94 | hsa-miR-326 | DDB2 | -0.43 | 0.26299 | 0.67 | 0.00378 | miRanda | -0.18 | 4.0E-5 | NA | |
| 95 | hsa-miR-106a-5p | FAS | -0.17 | 0.64287 | 0.66 | 0.0207 | miRNAWalker2 validate; miRTarBase | -0.16 | 0.00429 | 22431000; 27142596 | miR 106a is frequently upregulated in gastric cancer and inhibits the extrinsic apoptotic pathway by targeting FAS; Bioinformatic analysis combining with validation experiments identified FAS as a direct target of miR-106a; Moreover a significant inverse correlation was found between miR-106a and FAS expression not only in gastric cancer cell lines but also in gastric cancer specimens; Taken together these findings suggest that ectopicly overexpressed miR-106a may play an oncogenic role in gastric carcinogenesis and impair extrinsic apoptotic pathway through targeting FAS;Functional experiment ascertained that miR-106a interacted with FAS and mediated caspase3 pathway |
| 96 | hsa-miR-129-5p | FAS | -2.57 | 0 | 0.66 | 0.0207 | miRanda | -0.12 | 0.0007 | NA | |
| 97 | hsa-miR-148b-3p | GADD45A | 0.3 | 0.17466 | -0.74 | 0.00247 | MirTarget | -0.21 | 0.00928 | NA | |
| 98 | hsa-miR-454-3p | GADD45A | 0.63 | 0.01349 | -0.74 | 0.00247 | MirTarget; miRNATAP | -0.22 | 0.00181 | NA | |
| 99 | hsa-miR-1976 | GADD45B | 0.92 | 0.00012 | -1.92 | 0 | miRNATAP | -0.59 | 0 | NA | |
| 100 | hsa-miR-324-3p | GADD45B | 1.05 | 0.00039 | -1.92 | 0 | MirTarget; miRNATAP | -0.37 | 2.0E-5 | NA | |
| 101 | hsa-miR-590-3p | GADD45B | 1.12 | 0.00016 | -1.92 | 0 | miRanda | -0.37 | 2.0E-5 | NA | |
| 102 | hsa-miR-296-5p | GADD45G | 0.93 | 0.06024 | -1.31 | 0.00093 | miRNATAP | -0.17 | 0.00457 | NA | |
| 103 | hsa-miR-139-5p | GTSE1 | -1.83 | 0 | 3.35 | 0 | miRanda | -0.46 | 0 | NA | |
| 104 | hsa-let-7a-3p | IGF1 | 0.5 | 0.04111 | -2.08 | 0.00135 | mirMAP | -0.55 | 0.00422 | NA | |
| 105 | hsa-let-7b-3p | IGF1 | 0.22 | 0.29604 | -2.08 | 0.00135 | mirMAP | -0.64 | 0.00549 | NA | |
| 106 | hsa-let-7f-1-3p | IGF1 | 1.29 | 0 | -2.08 | 0.00135 | mirMAP | -0.85 | 0 | NA | |
| 107 | hsa-miR-103a-2-5p | IGF1 | 0.81 | 0.01999 | -2.08 | 0.00135 | mirMAP | -0.55 | 3.0E-5 | NA | |
| 108 | hsa-miR-130b-3p | IGF1 | 1.33 | 5.0E-5 | -2.08 | 0.00135 | MirTarget | -0.9 | 0 | NA | |
| 109 | hsa-miR-15b-3p | IGF1 | 1.76 | 0 | -2.08 | 0.00135 | mirMAP | -0.97 | 0 | NA | |
| 110 | hsa-miR-16-1-3p | IGF1 | 1.43 | 0 | -2.08 | 0.00135 | mirMAP | -0.89 | 0 | NA | |
| 111 | hsa-miR-181b-5p | IGF1 | 1.64 | 0 | -2.08 | 0.00135 | mirMAP | -0.48 | 0.00175 | NA | |
| 112 | hsa-miR-186-5p | IGF1 | 0.15 | 0.43471 | -2.08 | 0.00135 | mirMAP | -0.85 | 0.00061 | NA | |
| 113 | hsa-miR-19a-3p | IGF1 | 1.27 | 0.00011 | -2.08 | 0.00135 | MirTarget | -0.77 | 0 | NA | |
| 114 | hsa-miR-19b-1-5p | IGF1 | 1.39 | 0 | -2.08 | 0.00135 | mirMAP | -0.93 | 0 | NA | |
| 115 | hsa-miR-19b-3p | IGF1 | 0.76 | 0.00653 | -2.08 | 0.00135 | MirTarget | -0.82 | 0 | NA | |
| 116 | hsa-miR-20a-3p | IGF1 | 1.14 | 0.00045 | -2.08 | 0.00135 | mirMAP | -0.99 | 0 | NA | |
| 117 | hsa-miR-27a-3p | IGF1 | 1.3 | 0 | -2.08 | 0.00135 | miRNAWalker2 validate; miRTarBase | -0.62 | 0.00158 | NA | |
| 118 | hsa-miR-301a-3p | IGF1 | 1.45 | 1.0E-5 | -2.08 | 0.00135 | MirTarget | -0.44 | 0.00199 | NA | |
| 119 | hsa-miR-301b-3p | IGF1 | 1.1 | 0.07156 | -2.08 | 0.00135 | MirTarget | -0.49 | 0 | NA | |
| 120 | hsa-miR-3065-5p | IGF1 | -0.24 | 0.63312 | -2.08 | 0.00135 | mirMAP | -0.28 | 0.00304 | NA | |
| 121 | hsa-miR-32-3p | IGF1 | 0.58 | 0.11837 | -2.08 | 0.00135 | mirMAP | -0.53 | 0.00017 | NA | |
| 122 | hsa-miR-320a | IGF1 | 0.59 | 0.0119 | -2.08 | 0.00135 | miRNATAP | -0.57 | 0.00471 | NA | |
| 123 | hsa-miR-320b | IGF1 | 1.11 | 0.0005 | -2.08 | 0.00135 | miRNATAP | -0.64 | 1.0E-5 | NA | |
| 124 | hsa-miR-320c | IGF1 | 0.46 | 0.24061 | -2.08 | 0.00135 | miRNATAP | -0.42 | 0.00294 | NA | |
| 125 | hsa-miR-33a-3p | IGF1 | 0.35 | 0.32171 | -2.08 | 0.00135 | MirTarget | -0.68 | 0 | NA | |
| 126 | hsa-miR-3662 | IGF1 | 1.83 | 0.0054 | -2.08 | 0.00135 | MirTarget | -0.45 | 0 | NA | |
| 127 | hsa-miR-421 | IGF1 | 1.81 | 0 | -2.08 | 0.00135 | PITA | -0.45 | 0.00067 | NA | |
| 128 | hsa-miR-454-3p | IGF1 | 0.63 | 0.01349 | -2.08 | 0.00135 | MirTarget | -0.55 | 0.00337 | NA | |
| 129 | hsa-miR-486-5p | IGF1 | -0.63 | 0.15527 | -2.08 | 0.00135 | PITA; miRNATAP | -0.38 | 0.00034 | NA | |
| 130 | hsa-miR-576-5p | IGF1 | 0.63 | 0.04171 | -2.08 | 0.00135 | PITA; mirMAP; miRNATAP | -0.76 | 0 | NA | |
| 131 | hsa-miR-577 | IGF1 | 0.91 | 0.22561 | -2.08 | 0.00135 | PITA | -0.36 | 0 | NA | |
| 132 | hsa-miR-590-3p | IGF1 | 1.12 | 0.00016 | -2.08 | 0.00135 | MirTarget; miRanda; mirMAP; miRNATAP | -0.61 | 8.0E-5 | NA | |
| 133 | hsa-miR-592 | IGF1 | 0.78 | 0.2045 | -2.08 | 0.00135 | mirMAP | -0.41 | 0 | NA | |
| 134 | hsa-miR-629-5p | IGF1 | 1.06 | 0.00054 | -2.08 | 0.00135 | mirMAP | -0.88 | 0 | NA | |
| 135 | hsa-miR-940 | IGF1 | 1.64 | 0.00069 | -2.08 | 0.00135 | MirTarget; PITA; miRNATAP | -0.45 | 5.0E-5 | NA | |
| 136 | hsa-miR-143-3p | MDM2 | -0.66 | 0.04832 | 0.1 | 0.72515 | miRNAWalker2 validate | -0.19 | 0.00073 | NA | |
| 137 | hsa-let-7b-5p | MDM4 | 0.06 | 0.7814 | -0.43 | 0.0212 | miRNAWalker2 validate; MirTarget | -0.16 | 0.00995 | NA | |
| 138 | hsa-miR-127-5p | MDM4 | 0.68 | 0.02163 | -0.43 | 0.0212 | mirMAP | -0.13 | 0.0034 | NA | |
| 139 | hsa-miR-152-3p | MDM4 | 0.44 | 0.1617 | -0.43 | 0.0212 | MirTarget | -0.2 | 0 | NA | |
| 140 | hsa-miR-22-5p | MDM4 | 0.8 | 0.0008 | -0.43 | 0.0212 | MirTarget | -0.17 | 0.00197 | NA | |
| 141 | hsa-miR-27a-3p | MDM4 | 1.3 | 0 | -0.43 | 0.0212 | miRNATAP | -0.32 | 0 | NA | |
| 142 | hsa-miR-27b-3p | MDM4 | 0.08 | 0.72527 | -0.43 | 0.0212 | miRNATAP | -0.22 | 5.0E-5 | NA | |
| 143 | hsa-miR-126-5p | PERP | 0.42 | 0.07532 | 1.62 | 0.00158 | mirMAP | -0.42 | 0.00796 | NA | |
| 144 | hsa-miR-150-5p | PERP | 0.15 | 0.75372 | 1.62 | 0.00158 | MirTarget | -0.22 | 0.00462 | NA | |
| 145 | hsa-miR-192-5p | PERP | 0.08 | 0.94106 | 1.62 | 0.00158 | miRNAWalker2 validate | -0.28 | 0 | NA | |
| 146 | hsa-miR-215-5p | PERP | 0.72 | 0.57378 | 1.62 | 0.00158 | miRNAWalker2 validate | -0.22 | 0 | NA | |
| 147 | hsa-miR-29b-1-5p | PERP | 0.61 | 0.11636 | 1.62 | 0.00158 | MirTarget | -0.25 | 0.00925 | NA | |
| 148 | hsa-miR-362-3p | PERP | -0.03 | 0.91378 | 1.62 | 0.00158 | miRanda | -0.46 | 6.0E-5 | NA | |
| 149 | hsa-miR-577 | PERP | 0.91 | 0.22561 | 1.62 | 0.00158 | PITA | -0.22 | 1.0E-5 | NA | |
| 150 | hsa-miR-660-5p | PERP | -0.02 | 0.93887 | 1.62 | 0.00158 | MirTarget | -0.92 | 0 | NA |
| Num | GO | Overlap | Size | P Value | Adj. P Value |
|---|---|---|---|---|---|
| 1 | REGULATION OF CELL CYCLE | 27 | 949 | 1.089e-23 | 5.069e-20 |
| 2 | CELL CYCLE | 29 | 1316 | 1.362e-22 | 3.168e-19 |
| 3 | CELL CYCLE PROCESS | 25 | 1081 | 1.496e-19 | 2.32e-16 |
| 4 | REGULATION OF CELL DEATH | 27 | 1472 | 9.966e-19 | 1.159e-15 |
| 5 | POSITIVE REGULATION OF CELL DEATH | 20 | 605 | 2.294e-18 | 2.135e-15 |
| 6 | NEGATIVE REGULATION OF CELL CYCLE | 18 | 433 | 3.098e-18 | 2.402e-15 |
| 7 | MITOTIC CELL CYCLE | 21 | 766 | 1.136e-17 | 7.551e-15 |
| 8 | RESPONSE TO ABIOTIC STIMULUS | 23 | 1024 | 1.47e-17 | 7.601e-15 |
| 9 | CELL CYCLE CHECKPOINT | 14 | 194 | 1.468e-17 | 7.601e-15 |
| 10 | REGULATION OF CELL CYCLE ARREST | 12 | 108 | 1.942e-17 | 9.038e-15 |
| 11 | POSITIVE REGULATION OF PROTEIN METABOLIC PROCESS | 26 | 1492 | 2.192e-17 | 9.271e-15 |
| 12 | POSITIVE REGULATION OF CELL CYCLE | 16 | 332 | 3.233e-17 | 1.254e-14 |
| 13 | CELLULAR RESPONSE TO STRESS | 26 | 1565 | 7.054e-17 | 2.344e-14 |
| 14 | CELLULAR RESPONSE TO DNA DAMAGE STIMULUS | 20 | 720 | 6.694e-17 | 2.344e-14 |
| 15 | CELL DEATH | 22 | 1001 | 1.537e-16 | 4.47e-14 |
| 16 | SIGNAL TRANSDUCTION BY P53 CLASS MEDIATOR | 12 | 127 | 1.451e-16 | 4.47e-14 |
| 17 | POSITIVE REGULATION OF CELL CYCLE PROCESS | 14 | 247 | 4.407e-16 | 1.139e-13 |
| 18 | NEGATIVE REGULATION OF CELL CYCLE G1 S PHASE TRANSITION | 11 | 98 | 4.267e-16 | 1.139e-13 |
| 19 | DNA INTEGRITY CHECKPOINT | 12 | 146 | 8.046e-16 | 1.97e-13 |
| 20 | G1 DNA DAMAGE CHECKPOINT | 10 | 73 | 1.294e-15 | 3.011e-13 |
| 21 | NEGATIVE REGULATION OF CELL CYCLE PROCESS | 13 | 214 | 2.317e-15 | 5.135e-13 |
| 22 | POSITIVE REGULATION OF CELL CYCLE ARREST | 10 | 85 | 6.384e-15 | 1.35e-12 |
| 23 | REGULATION OF MITOTIC CELL CYCLE | 16 | 468 | 7.107e-15 | 1.438e-12 |
| 24 | REGULATION OF PROTEOLYSIS | 18 | 711 | 1.791e-14 | 3.473e-12 |
| 25 | ACTIVATION OF CYSTEINE TYPE ENDOPEPTIDASE ACTIVITY | 10 | 95 | 2.026e-14 | 3.771e-12 |
| 26 | SIGNAL TRANSDUCTION IN RESPONSE TO DNA DAMAGE | 10 | 96 | 2.258e-14 | 4.041e-12 |
| 27 | AGING | 13 | 264 | 3.515e-14 | 5.452e-12 |
| 28 | NEGATIVE REGULATION OF MITOTIC CELL CYCLE | 12 | 199 | 3.434e-14 | 5.452e-12 |
| 29 | MITOTIC DNA INTEGRITY CHECKPOINT | 10 | 100 | 3.441e-14 | 5.452e-12 |
| 30 | CELLULAR RESPONSE TO ABIOTIC STIMULUS | 13 | 263 | 3.347e-14 | 5.452e-12 |
| 31 | NEGATIVE REGULATION OF CELL CYCLE PHASE TRANSITION | 11 | 146 | 3.84e-14 | 5.763e-12 |
| 32 | REGULATION OF CELL CYCLE G1 S PHASE TRANSITION | 11 | 147 | 4.143e-14 | 6.024e-12 |
| 33 | INTRINSIC APOPTOTIC SIGNALING PATHWAY | 11 | 152 | 6.012e-14 | 8.477e-12 |
| 34 | REGULATION OF CYSTEINE TYPE ENDOPEPTIDASE ACTIVITY | 12 | 213 | 7.759e-14 | 1.062e-11 |
| 35 | POSITIVE REGULATION OF PROTEOLYSIS | 14 | 363 | 9.053e-14 | 1.204e-11 |
| 36 | APOPTOTIC SIGNALING PATHWAY | 13 | 289 | 1.123e-13 | 1.412e-11 |
| 37 | ZYMOGEN ACTIVATION | 10 | 112 | 1.102e-13 | 1.412e-11 |
| 38 | REGULATION OF TRANSFERASE ACTIVITY | 19 | 946 | 1.747e-13 | 2.139e-11 |
| 39 | REGULATION OF PEPTIDASE ACTIVITY | 14 | 392 | 2.583e-13 | 3.081e-11 |
| 40 | RESPONSE TO OXYGEN LEVELS | 13 | 311 | 2.867e-13 | 3.334e-11 |
| 41 | RESPONSE TO UV | 10 | 126 | 3.661e-13 | 4.154e-11 |
| 42 | REGULATION OF CELL CYCLE PHASE TRANSITION | 13 | 321 | 4.289e-13 | 4.752e-11 |
| 43 | CELL CYCLE PHASE TRANSITION | 12 | 255 | 6.608e-13 | 7.151e-11 |
| 44 | CELLULAR RESPONSE TO RADIATION | 10 | 137 | 8.548e-13 | 9.039e-11 |
| 45 | REGULATION OF PROTEIN MODIFICATION PROCESS | 23 | 1710 | 8.953e-13 | 9.258e-11 |
| 46 | MITOTIC CELL CYCLE CHECKPOINT | 10 | 139 | 9.896e-13 | 1.001e-10 |
| 47 | REGULATION OF KINASE ACTIVITY | 17 | 776 | 1.183e-12 | 1.171e-10 |
| 48 | REGULATION OF CYCLIN DEPENDENT PROTEIN KINASE ACTIVITY | 9 | 97 | 1.458e-12 | 1.413e-10 |
| 49 | REGULATION OF CELL CYCLE PROCESS | 15 | 558 | 1.876e-12 | 1.781e-10 |
| 50 | REGULATION OF APOPTOTIC SIGNALING PATHWAY | 13 | 363 | 2.037e-12 | 1.896e-10 |
| 51 | POSITIVE REGULATION OF PEPTIDASE ACTIVITY | 10 | 154 | 2.777e-12 | 2.533e-10 |
| 52 | REGULATION OF PROTEIN SERINE THREONINE KINASE ACTIVITY | 14 | 470 | 3.005e-12 | 2.689e-10 |
| 53 | CELLULAR RESPONSE TO UV | 8 | 66 | 3.083e-12 | 2.706e-10 |
| 54 | REGULATION OF RESPONSE TO STRESS | 21 | 1468 | 4.281e-12 | 3.689e-10 |
| 55 | POSITIVE REGULATION OF APOPTOTIC SIGNALING PATHWAY | 10 | 171 | 7.917e-12 | 6.698e-10 |
| 56 | POSITIVE REGULATION OF CATALYTIC ACTIVITY | 21 | 1518 | 8.112e-12 | 6.74e-10 |
| 57 | RESPONSE TO RADIATION | 13 | 413 | 1.031e-11 | 8.417e-10 |
| 58 | INTRACELLULAR SIGNAL TRANSDUCTION | 21 | 1572 | 1.576e-11 | 1.264e-09 |
| 59 | POSITIVE REGULATION OF MOLECULAR FUNCTION | 22 | 1791 | 2.159e-11 | 1.703e-09 |
| 60 | REGULATION OF PHOSPHORUS METABOLIC PROCESS | 21 | 1618 | 2.719e-11 | 2.109e-09 |
| 61 | REGULATION OF CELLULAR RESPONSE TO STRESS | 15 | 691 | 3.891e-11 | 2.968e-09 |
| 62 | CELLULAR RESPONSE TO LIGHT STIMULUS | 8 | 91 | 4.363e-11 | 3.274e-09 |
| 63 | REPLICATIVE SENESCENCE | 5 | 12 | 5.025e-11 | 3.711e-09 |
| 64 | REGULATION OF CELL PROLIFERATION | 20 | 1496 | 5.82e-11 | 4.232e-09 |
| 65 | CELL CYCLE ARREST | 9 | 154 | 9.76e-11 | 6.881e-09 |
| 66 | POSITIVE REGULATION OF INTRACELLULAR SIGNAL TRANSDUCTION | 16 | 876 | 9.679e-11 | 6.881e-09 |
| 67 | REGULATION OF SIGNAL TRANSDUCTION BY P53 CLASS MEDIATOR | 9 | 162 | 1.536e-10 | 1.067e-08 |
| 68 | CELL CYCLE G1 S PHASE TRANSITION | 8 | 111 | 2.187e-10 | 1.475e-08 |
| 69 | G1 S TRANSITION OF MITOTIC CELL CYCLE | 8 | 111 | 2.187e-10 | 1.475e-08 |
| 70 | RESPONSE TO DRUG | 12 | 431 | 2.976e-10 | 1.978e-08 |
| 71 | REGULATION OF INTRACELLULAR SIGNAL TRANSDUCTION | 20 | 1656 | 3.575e-10 | 2.343e-08 |
| 72 | POSITIVE REGULATION OF PROTEIN MODIFICATION PROCESS | 17 | 1135 | 4.64e-10 | 2.999e-08 |
| 73 | PROTEIN MATURATION | 10 | 265 | 5.913e-10 | 3.769e-08 |
| 74 | REGULATION OF FIBROBLAST PROLIFERATION | 7 | 81 | 8.835e-10 | 5.555e-08 |
| 75 | RESPONSE TO LIGHT STIMULUS | 10 | 280 | 1.008e-09 | 6.256e-08 |
| 76 | RESPONSE TO STEROID HORMONE | 12 | 497 | 1.502e-09 | 9.196e-08 |
| 77 | RESPONSE TO IONIZING RADIATION | 8 | 145 | 1.856e-09 | 1.122e-07 |
| 78 | REGULATION OF EXTRINSIC APOPTOTIC SIGNALING PATHWAY | 8 | 153 | 2.842e-09 | 1.695e-07 |
| 79 | INTRINSIC APOPTOTIC SIGNALING PATHWAY BY P53 CLASS MEDIATOR | 6 | 53 | 2.914e-09 | 1.716e-07 |
| 80 | REGULATION OF EXTRINSIC APOPTOTIC SIGNALING PATHWAY VIA DEATH DOMAIN RECEPTORS | 6 | 55 | 3.667e-09 | 2.133e-07 |
| 81 | RESPONSE TO METAL ION | 10 | 333 | 5.341e-09 | 3.068e-07 |
| 82 | REGULATION OF DNA DAMAGE RESPONSE SIGNAL TRANSDUCTION BY P53 CLASS MEDIATOR | 5 | 28 | 6.059e-09 | 3.438e-07 |
| 83 | CELL DIVISION | 11 | 460 | 8.876e-09 | 4.976e-07 |
| 84 | POSITIVE REGULATION OF PHOSPHATE METABOLIC PROCESS | 15 | 1036 | 1.024e-08 | 5.604e-07 |
| 85 | POSITIVE REGULATION OF PHOSPHORUS METABOLIC PROCESS | 15 | 1036 | 1.024e-08 | 5.604e-07 |
| 86 | RESPONSE TO KETONE | 8 | 182 | 1.115e-08 | 6.031e-07 |
| 87 | CELLULAR RESPONSE TO EXTERNAL STIMULUS | 9 | 264 | 1.136e-08 | 6.075e-07 |
| 88 | RESPONSE TO ALCOHOL | 10 | 362 | 1.183e-08 | 6.256e-07 |
| 89 | RESPONSE TO LIPID | 14 | 888 | 1.231e-08 | 6.386e-07 |
| 90 | CELL AGING | 6 | 67 | 1.235e-08 | 6.386e-07 |
| 91 | POSITIVE REGULATION OF TRANSFERASE ACTIVITY | 12 | 616 | 1.656e-08 | 8.466e-07 |
| 92 | DNA METABOLIC PROCESS | 13 | 758 | 1.7e-08 | 8.597e-07 |
| 93 | INTRINSIC APOPTOTIC SIGNALING PATHWAY IN RESPONSE TO DNA DAMAGE | 6 | 71 | 1.76e-08 | 8.805e-07 |
| 94 | RESPONSE TO ORGANIC CYCLIC COMPOUND | 14 | 917 | 1.846e-08 | 9.139e-07 |
| 95 | NEGATIVE REGULATION OF PROTEIN METABOLIC PROCESS | 15 | 1087 | 1.948e-08 | 9.539e-07 |
| 96 | NEURON APOPTOTIC PROCESS | 5 | 35 | 1.976e-08 | 9.579e-07 |
| 97 | POSITIVE REGULATION OF DNA DAMAGE RESPONSE SIGNAL TRANSDUCTION BY P53 CLASS MEDIATOR | 4 | 13 | 2.055e-08 | 9.856e-07 |
| 98 | POSITIVE REGULATION OF MITOCHONDRIAL OUTER MEMBRANE PERMEABILIZATION INVOLVED IN APOPTOTIC SIGNALING PATHWAY | 5 | 36 | 2.291e-08 | 1.088e-06 |
| 99 | NEGATIVE REGULATION OF CELL PROLIFERATION | 12 | 643 | 2.658e-08 | 1.249e-06 |
| 100 | DNA REPLICATION | 8 | 208 | 3.159e-08 | 1.47e-06 |
| 101 | POSITIVE REGULATION OF CELL COMMUNICATION | 17 | 1532 | 4.236e-08 | 1.952e-06 |
| 102 | RESPONSE TO ESTROGEN | 8 | 218 | 4.546e-08 | 2.074e-06 |
| 103 | CELLULAR RESPONSE TO OXYGEN LEVELS | 7 | 143 | 4.75e-08 | 2.146e-06 |
| 104 | REGULATION OF PROTEIN STABILITY | 8 | 221 | 5.053e-08 | 2.261e-06 |
| 105 | REGULATION OF RESPONSE TO DNA DAMAGE STIMULUS | 7 | 145 | 5.227e-08 | 2.316e-06 |
| 106 | REGULATION OF MITOCHONDRIAL OUTER MEMBRANE PERMEABILIZATION INVOLVED IN APOPTOTIC SIGNALING PATHWAY | 5 | 43 | 5.777e-08 | 2.536e-06 |
| 107 | RESPONSE TO OXYGEN CONTAINING COMPOUND | 16 | 1381 | 6.67e-08 | 2.901e-06 |
| 108 | POSITIVE REGULATION OF SIGNAL TRANSDUCTION BY P53 CLASS MEDIATOR | 4 | 17 | 6.791e-08 | 2.926e-06 |
| 109 | PROTEOLYSIS | 15 | 1208 | 7.871e-08 | 3.36e-06 |
| 110 | RESPONSE TO EXTERNAL STIMULUS | 18 | 1821 | 8.661e-08 | 3.664e-06 |
| 111 | NEURON DEATH | 5 | 47 | 9.14e-08 | 3.797e-06 |
| 112 | POSITIVE REGULATION OF NEURON APOPTOTIC PROCESS | 5 | 47 | 9.14e-08 | 3.797e-06 |
| 113 | RESPONSE TO TOXIC SUBSTANCE | 8 | 241 | 9.855e-08 | 4.058e-06 |
| 114 | REGENERATION | 7 | 161 | 1.072e-07 | 4.377e-06 |
| 115 | RESPONSE TO HORMONE | 13 | 893 | 1.159e-07 | 4.691e-06 |
| 116 | RESPONSE TO ENDOGENOUS STIMULUS | 16 | 1450 | 1.309e-07 | 5.249e-06 |
| 117 | NEGATIVE REGULATION OF CELL CYCLE ARREST | 4 | 20 | 1.375e-07 | 5.469e-06 |
| 118 | NEGATIVE REGULATION OF MOLECULAR FUNCTION | 14 | 1079 | 1.399e-07 | 5.515e-06 |
| 119 | POSITIVE REGULATION OF CELLULAR PROTEIN LOCALIZATION | 9 | 360 | 1.621e-07 | 6.339e-06 |
| 120 | RESPONSE TO INORGANIC SUBSTANCE | 10 | 479 | 1.636e-07 | 6.343e-06 |
| 121 | POSITIVE REGULATION OF FIBROBLAST PROLIFERATION | 5 | 53 | 1.692e-07 | 6.451e-06 |
| 122 | POSITIVE REGULATION OF EXTRINSIC APOPTOTIC SIGNALING PATHWAY | 5 | 53 | 1.692e-07 | 6.451e-06 |
| 123 | POSITIVE REGULATION OF KINASE ACTIVITY | 10 | 482 | 1.733e-07 | 6.556e-06 |
| 124 | RESPONSE TO CORTICOSTEROID | 7 | 176 | 1.97e-07 | 7.392e-06 |
| 125 | POSITIVE REGULATION OF RESPONSE TO STIMULUS | 18 | 1929 | 2.077e-07 | 7.672e-06 |
| 126 | POSITIVE REGULATION OF PROTEIN OLIGOMERIZATION | 4 | 22 | 2.069e-07 | 7.672e-06 |
| 127 | REGULATION OF CELL CYCLE G2 M PHASE TRANSITION | 5 | 59 | 2.919e-07 | 1.07e-05 |
| 128 | POSITIVE REGULATION OF PROTEIN SERINE THREONINE KINASE ACTIVITY | 8 | 289 | 3.944e-07 | 1.434e-05 |
| 129 | NEGATIVE REGULATION OF CATALYTIC ACTIVITY | 12 | 829 | 4.177e-07 | 1.503e-05 |
| 130 | RESPONSE TO CORTICOSTERONE | 4 | 26 | 4.199e-07 | 1.503e-05 |
| 131 | NEGATIVE REGULATION OF APOPTOTIC SIGNALING PATHWAY | 7 | 200 | 4.684e-07 | 1.664e-05 |
| 132 | POSITIVE REGULATION OF MITOTIC CELL CYCLE | 6 | 123 | 4.754e-07 | 1.676e-05 |
| 133 | POSITIVE REGULATION OF NEURON DEATH | 5 | 67 | 5.552e-07 | 1.942e-05 |
| 134 | POSITIVE REGULATION OF LEUKOCYTE APOPTOTIC PROCESS | 4 | 28 | 5.73e-07 | 1.99e-05 |
| 135 | REGULATION OF CELLULAR PROTEIN LOCALIZATION | 10 | 552 | 6.019e-07 | 2.075e-05 |
| 136 | RESPONSE TO NITROGEN COMPOUND | 12 | 859 | 6.096e-07 | 2.086e-05 |
| 137 | REGULATION OF PROTEIN INSERTION INTO MITOCHONDRIAL MEMBRANE INVOLVED IN APOPTOTIC SIGNALING PATHWAY | 4 | 29 | 6.636e-07 | 2.237e-05 |
| 138 | POSITIVE REGULATION OF PROTEIN INSERTION INTO MITOCHONDRIAL MEMBRANE INVOLVED IN APOPTOTIC SIGNALING PATHWAY | 4 | 29 | 6.636e-07 | 2.237e-05 |
| 139 | PROTEIN STABILIZATION | 6 | 131 | 6.892e-07 | 2.3e-05 |
| 140 | REGULATION OF MEMBRANE PERMEABILITY | 5 | 70 | 6.92e-07 | 2.3e-05 |
| 141 | NEGATIVE REGULATION OF CELL DEATH | 12 | 872 | 7.147e-07 | 2.358e-05 |
| 142 | NEGATIVE REGULATION OF CELL MATRIX ADHESION | 4 | 30 | 7.643e-07 | 2.487e-05 |
| 143 | RESPONSE TO X RAY | 4 | 30 | 7.643e-07 | 2.487e-05 |
| 144 | REGULATION OF MITOCHONDRION ORGANIZATION | 7 | 218 | 8.364e-07 | 2.702e-05 |
| 145 | POSITIVE REGULATION OF ORGANELLE ORGANIZATION | 10 | 573 | 8.452e-07 | 2.712e-05 |
| 146 | RESPONSE TO EXTRACELLULAR STIMULUS | 9 | 441 | 8.886e-07 | 2.832e-05 |
| 147 | INTRINSIC APOPTOTIC SIGNALING PATHWAY IN RESPONSE TO ENDOPLASMIC RETICULUM STRESS | 4 | 32 | 9.994e-07 | 3.163e-05 |
| 148 | REGULATION OF LEUKOCYTE APOPTOTIC PROCESS | 5 | 79 | 1.268e-06 | 3.986e-05 |
| 149 | RESPONSE TO ESTRADIOL | 6 | 146 | 1.301e-06 | 4.064e-05 |
| 150 | REGULATION OF DNA METABOLIC PROCESS | 8 | 340 | 1.341e-06 | 4.159e-05 |
| 151 | RESPONSE TO TRANSITION METAL NANOPARTICLE | 6 | 148 | 1.409e-06 | 4.341e-05 |
| 152 | REGULATION OF PROTEIN OLIGOMERIZATION | 4 | 35 | 1.447e-06 | 4.402e-05 |
| 153 | RESPONSE TO MINERALOCORTICOID | 4 | 35 | 1.447e-06 | 4.402e-05 |
| 154 | ORGAN REGENERATION | 5 | 83 | 1.622e-06 | 4.88e-05 |
| 155 | POSITIVE REGULATION OF CYCLIN DEPENDENT PROTEIN KINASE ACTIVITY | 4 | 36 | 1.626e-06 | 4.88e-05 |
| 156 | REGULATION OF HYDROLASE ACTIVITY | 14 | 1327 | 1.703e-06 | 5.079e-05 |
| 157 | REGULATION OF INTRACELLULAR TRANSPORT | 10 | 621 | 1.747e-06 | 5.178e-05 |
| 158 | POSITIVE REGULATION OF CELLULAR COMPONENT ORGANIZATION | 13 | 1152 | 2.091e-06 | 6.159e-05 |
| 159 | ACTIVATION OF MAPKKK ACTIVITY | 3 | 11 | 2.112e-06 | 6.181e-05 |
| 160 | REGULATION OF NEURON DEATH | 7 | 252 | 2.199e-06 | 6.394e-05 |
| 161 | REGULATION OF CELL MATRIX ADHESION | 5 | 90 | 2.424e-06 | 7.006e-05 |
| 162 | POSITIVE REGULATION OF INTRACELLULAR TRANSPORT | 8 | 370 | 2.516e-06 | 7.226e-05 |
| 163 | MITOCHONDRIAL MEMBRANE ORGANIZATION | 5 | 92 | 2.703e-06 | 7.716e-05 |
| 164 | ACTIVATION OF CYSTEINE TYPE ENDOPEPTIDASE ACTIVITY INVOLVED IN APOPTOTIC SIGNALING PATHWAY | 3 | 13 | 3.648e-06 | 0.0001029 |
| 165 | RESPONSE TO COBALT ION | 3 | 13 | 3.648e-06 | 0.0001029 |
| 166 | NEGATIVE REGULATION OF EXTRINSIC APOPTOTIC SIGNALING PATHWAY | 5 | 98 | 3.693e-06 | 0.0001029 |
| 167 | REGULATION OF RELEASE OF CYTOCHROME C FROM MITOCHONDRIA | 4 | 44 | 3.694e-06 | 0.0001029 |
| 168 | POSITIVE REGULATION OF P38MAPK CASCADE | 3 | 14 | 4.636e-06 | 0.0001284 |
| 169 | REGULATION OF EPITHELIAL CELL PROLIFERATION | 7 | 285 | 4.951e-06 | 0.0001363 |
| 170 | RESPONSE TO GAMMA RADIATION | 4 | 50 | 6.201e-06 | 0.0001687 |
| 171 | RESPONSE TO NUTRIENT | 6 | 191 | 6.165e-06 | 0.0001687 |
| 172 | REGULATION OF NEURON APOPTOTIC PROCESS | 6 | 192 | 6.353e-06 | 0.0001719 |
| 173 | NEGATIVE REGULATION OF PHOSPHORYLATION | 8 | 422 | 6.624e-06 | 0.0001782 |
| 174 | CELLULAR RESPONSE TO IONIZING RADIATION | 4 | 52 | 7.264e-06 | 0.0001931 |
| 175 | POSITIVE REGULATION OF INTRINSIC APOPTOTIC SIGNALING PATHWAY | 4 | 52 | 7.264e-06 | 0.0001931 |
| 176 | REGULATION OF CATABOLIC PROCESS | 10 | 731 | 7.431e-06 | 0.0001965 |
| 177 | NEGATIVE REGULATION OF CELL SUBSTRATE ADHESION | 4 | 53 | 7.843e-06 | 0.0002062 |
| 178 | REGULATION OF LYMPHOCYTE APOPTOTIC PROCESS | 4 | 54 | 8.455e-06 | 0.000221 |
| 179 | POSITIVE REGULATION OF CYSTEINE TYPE ENDOPEPTIDASE ACTIVITY INVOLVED IN APOPTOTIC SIGNALING PATHWAY | 3 | 17 | 8.616e-06 | 0.0002227 |
| 180 | POSITIVE REGULATION OF EXTRINSIC APOPTOTIC SIGNALING PATHWAY VIA DEATH DOMAIN RECEPTORS | 3 | 17 | 8.616e-06 | 0.0002227 |
| 181 | MITOCHONDRION ORGANIZATION | 9 | 594 | 1.009e-05 | 0.0002593 |
| 182 | POSITIVE REGULATION OF MITOCHONDRIAL MEMBRANE PERMEABILITY | 3 | 18 | 1.032e-05 | 0.0002639 |
| 183 | APOPTOTIC MITOCHONDRIAL CHANGES | 4 | 57 | 1.051e-05 | 0.0002671 |
| 184 | RESPONSE TO MECHANICAL STIMULUS | 6 | 210 | 1.06e-05 | 0.0002681 |
| 185 | DEVELOPMENTAL PROCESS INVOLVED IN REPRODUCTION | 9 | 602 | 1.123e-05 | 0.0002823 |
| 186 | NEGATIVE REGULATION OF PROTEOLYSIS | 7 | 329 | 1.262e-05 | 0.0003157 |
| 187 | NEGATIVE REGULATION OF RESPONSE TO STIMULUS | 13 | 1360 | 1.283e-05 | 0.0003191 |
| 188 | REGULATION OF ESTABLISHMENT OF PROTEIN LOCALIZATION TO MITOCHONDRION | 5 | 128 | 1.363e-05 | 0.0003374 |
| 189 | POSITIVE REGULATION OF LYMPHOCYTE APOPTOTIC PROCESS | 3 | 20 | 1.437e-05 | 0.0003538 |
| 190 | POSITIVE REGULATION OF MAPK CASCADE | 8 | 470 | 1.449e-05 | 0.0003548 |
| 191 | REGULATION OF ORGANELLE ORGANIZATION | 12 | 1178 | 1.589e-05 | 0.000387 |
| 192 | POSITIVE REGULATION OF RESPONSE TO DNA DAMAGE STIMULUS | 4 | 64 | 1.669e-05 | 0.0004045 |
| 193 | DNA REPAIR | 8 | 480 | 1.686e-05 | 0.0004065 |
| 194 | NEGATIVE REGULATION OF CELL COMMUNICATION | 12 | 1192 | 1.788e-05 | 0.0004288 |
| 195 | REGULATION OF MITOCHONDRIAL MEMBRANE PERMEABILITY INVOLVED IN APOPTOTIC PROCESS | 3 | 22 | 1.935e-05 | 0.0004524 |
| 196 | RESPONSE TO ENDOPLASMIC RETICULUM STRESS | 6 | 233 | 1.911e-05 | 0.0004524 |
| 197 | RELEASE OF CYTOCHROME C FROM MITOCHONDRIA | 3 | 22 | 1.935e-05 | 0.0004524 |
| 198 | NEGATIVE REGULATION OF TRANSFERASE ACTIVITY | 7 | 351 | 1.916e-05 | 0.0004524 |
| 199 | REGULATION OF CYSTEINE TYPE ENDOPEPTIDASE ACTIVITY INVOLVED IN APOPTOTIC SIGNALING PATHWAY | 3 | 22 | 1.935e-05 | 0.0004524 |
| 200 | CELL CYCLE G2 M PHASE TRANSITION | 5 | 138 | 1.962e-05 | 0.0004564 |
| 201 | POSITIVE REGULATION OF CELL CYCLE PHASE TRANSITION | 4 | 68 | 2.124e-05 | 0.0004918 |
| 202 | RESPONSE TO MAGNESIUM ION | 3 | 23 | 2.221e-05 | 0.0005042 |
| 203 | RESPONSE TO INCREASED OXYGEN LEVELS | 3 | 23 | 2.221e-05 | 0.0005042 |
| 204 | RESPONSE TO HYPEROXIA | 3 | 23 | 2.221e-05 | 0.0005042 |
| 205 | PROTEIN INSERTION INTO MEMBRANE | 3 | 23 | 2.221e-05 | 0.0005042 |
| 206 | REGULATION OF INTRINSIC APOPTOTIC SIGNALING PATHWAY | 5 | 145 | 2.49e-05 | 0.0005625 |
| 207 | REGULATION OF EXECUTION PHASE OF APOPTOSIS | 3 | 24 | 2.534e-05 | 0.0005697 |
| 208 | REGULATION OF PROTEASOMAL UBIQUITIN DEPENDENT PROTEIN CATABOLIC PROCESS | 5 | 148 | 2.748e-05 | 0.0006147 |
| 209 | NEGATIVE REGULATION OF KINASE ACTIVITY | 6 | 250 | 2.84e-05 | 0.0006323 |
| 210 | HISTONE PHOSPHORYLATION | 3 | 25 | 2.875e-05 | 0.0006371 |
| 211 | REGULATION OF P38MAPK CASCADE | 3 | 26 | 3.245e-05 | 0.0007155 |
| 212 | REGULATION OF CELLULAR LOCALIZATION | 12 | 1277 | 3.539e-05 | 0.0007767 |
| 213 | NEGATIVE REGULATION OF PHOSPHORUS METABOLIC PROCESS | 8 | 541 | 3.96e-05 | 0.0008571 |
| 214 | REGULATION OF PROTEIN CATABOLIC PROCESS | 7 | 393 | 3.943e-05 | 0.0008571 |
| 215 | NEGATIVE REGULATION OF PHOSPHATE METABOLIC PROCESS | 8 | 541 | 3.96e-05 | 0.0008571 |
| 216 | CELLULAR RESPONSE TO MECHANICAL STIMULUS | 4 | 80 | 4.04e-05 | 0.0008702 |
| 217 | POSITIVE REGULATION OF RELEASE OF CYTOCHROME C FROM MITOCHONDRIA | 3 | 28 | 4.075e-05 | 0.0008737 |
| 218 | REGULATION OF NUCLEAR DIVISION | 5 | 163 | 4.362e-05 | 0.0009311 |
| 219 | POSITIVE REGULATION OF HYDROLASE ACTIVITY | 10 | 905 | 4.674e-05 | 0.0009931 |
| 220 | REGULATION OF CELLULAR PROTEIN CATABOLIC PROCESS | 6 | 274 | 4.739e-05 | 0.001002 |
| 221 | POSITIVE REGULATION OF CELLULAR COMPONENT BIOGENESIS | 7 | 406 | 4.845e-05 | 0.00102 |
| 222 | POSITIVE REGULATION OF MITOCHONDRION ORGANIZATION | 5 | 167 | 4.897e-05 | 0.001026 |
| 223 | INTRINSIC APOPTOTIC SIGNALING PATHWAY IN RESPONSE TO DNA DAMAGE BY P53 CLASS MEDIATOR | 3 | 30 | 5.033e-05 | 0.001037 |
| 224 | NEGATIVE REGULATION OF B CELL ACTIVATION | 3 | 30 | 5.033e-05 | 0.001037 |
| 225 | REPRODUCTIVE SYSTEM DEVELOPMENT | 7 | 408 | 4.998e-05 | 0.001037 |
| 226 | RESPONSE TO CARBOHYDRATE | 5 | 168 | 5.038e-05 | 0.001037 |
| 227 | REGULATION OF CELL SUBSTRATE ADHESION | 5 | 173 | 5.791e-05 | 0.001187 |
| 228 | NEGATIVE REGULATION OF CYCLIN DEPENDENT PROTEIN KINASE ACTIVITY | 3 | 32 | 6.128e-05 | 0.001251 |
| 229 | PROTEIN CATABOLIC PROCESS | 8 | 579 | 6.388e-05 | 0.001298 |
| 230 | MITOCHONDRIAL TRANSPORT | 5 | 177 | 6.455e-05 | 0.001306 |
| 231 | REGULATION OF PROTEIN EXPORT FROM NUCLEUS | 3 | 33 | 6.729e-05 | 0.001338 |
| 232 | SIGNAL TRANSDUCTION IN ABSENCE OF LIGAND | 3 | 33 | 6.729e-05 | 0.001338 |
| 233 | EXTRINSIC APOPTOTIC SIGNALING PATHWAY IN ABSENCE OF LIGAND | 3 | 33 | 6.729e-05 | 0.001338 |
| 234 | REGULATION OF CELL AGING | 3 | 33 | 6.729e-05 | 0.001338 |
| 235 | REGULATION OF PROTEIN LOCALIZATION | 10 | 950 | 7.027e-05 | 0.001391 |
| 236 | REGULATION OF PROTEASOMAL PROTEIN CATABOLIC PROCESS | 5 | 181 | 7.176e-05 | 0.001415 |
| 237 | T CELL HOMEOSTASIS | 3 | 34 | 7.368e-05 | 0.001428 |
| 238 | PROTEIN DESTABILIZATION | 3 | 34 | 7.368e-05 | 0.001428 |
| 239 | PROTEIN KINASE B SIGNALING | 3 | 34 | 7.368e-05 | 0.001428 |
| 240 | NEGATIVE REGULATION OF EXTRINSIC APOPTOTIC SIGNALING PATHWAY VIA DEATH DOMAIN RECEPTORS | 3 | 34 | 7.368e-05 | 0.001428 |
| 241 | REGULATION OF CELLULAR COMPONENT BIOGENESIS | 9 | 767 | 7.491e-05 | 0.001446 |
| 242 | RHYTHMIC PROCESS | 6 | 298 | 7.545e-05 | 0.001451 |
| 243 | NEGATIVE REGULATION OF INTRACELLULAR SIGNAL TRANSDUCTION | 7 | 437 | 7.696e-05 | 0.001474 |
| 244 | NEGATIVE REGULATION OF PROTEIN PROCESSING | 3 | 35 | 8.045e-05 | 0.001516 |
| 245 | NEGATIVE REGULATION OF PROTEIN MATURATION | 3 | 35 | 8.045e-05 | 0.001516 |
| 246 | RESPONSE TO IRON ION | 3 | 35 | 8.045e-05 | 0.001516 |
| 247 | REGULATION OF RESPONSE TO REACTIVE OXYGEN SPECIES | 3 | 35 | 8.045e-05 | 0.001516 |
| 248 | CELLULAR RESPONSE TO EXTRACELLULAR STIMULUS | 5 | 188 | 8.585e-05 | 0.001611 |
| 249 | EXTRINSIC APOPTOTIC SIGNALING PATHWAY | 4 | 99 | 9.299e-05 | 0.001738 |
| 250 | NEGATIVE REGULATION OF PROTEIN MODIFICATION PROCESS | 8 | 616 | 9.838e-05 | 0.001831 |
| 251 | CELLULAR RESPONSE TO LIPID | 7 | 457 | 0.0001018 | 0.001886 |
| 252 | POSITIVE REGULATION OF PROTEIN COMPLEX ASSEMBLY | 5 | 197 | 0.000107 | 0.001975 |
| 253 | EXTRINSIC APOPTOTIC SIGNALING PATHWAY VIA DEATH DOMAIN RECEPTORS | 3 | 39 | 0.0001116 | 0.002052 |
| 254 | CELLULAR RESPONSE TO ORGANIC CYCLIC COMPOUND | 7 | 465 | 0.0001133 | 0.002076 |
| 255 | REGULATION OF GROWTH | 8 | 633 | 0.0001188 | 0.002167 |
| 256 | CELLULAR RESPONSE TO ESTROGEN STIMULUS | 3 | 41 | 0.0001297 | 0.002358 |
| 257 | PHOSPHATE CONTAINING COMPOUND METABOLIC PROCESS | 14 | 1977 | 0.0001537 | 0.002782 |
| 258 | REGULATION OF MAPK CASCADE | 8 | 660 | 0.0001583 | 0.002855 |
| 259 | LYMPHOCYTE ACTIVATION | 6 | 342 | 0.0001603 | 0.00288 |
| 260 | CELLULAR RESPONSE TO STEROID HORMONE STIMULUS | 5 | 218 | 0.0001718 | 0.003075 |
| 261 | REGULATION OF NUCLEOCYTOPLASMIC TRANSPORT | 5 | 220 | 0.0001793 | 0.003197 |
| 262 | RESPONSE TO ANTIBIOTIC | 3 | 47 | 0.0001953 | 0.003469 |
| 263 | REPRODUCTION | 11 | 1297 | 0.0001991 | 0.003522 |
| 264 | REGULATION OF B CELL ACTIVATION | 4 | 121 | 0.0002019 | 0.003559 |
| 265 | POSITIVE REGULATION OF ESTABLISHMENT OF PROTEIN LOCALIZATION | 7 | 514 | 0.0002101 | 0.003689 |
| 266 | RESPONSE TO BIOTIC STIMULUS | 9 | 886 | 0.000223 | 0.003901 |
| 267 | LYMPHOCYTE HOMEOSTASIS | 3 | 50 | 0.0002349 | 0.004094 |
| 268 | NEGATIVE REGULATION OF PROTEIN SERINE THREONINE KINASE ACTIVITY | 4 | 126 | 0.0002358 | 0.004094 |
| 269 | REGULATION OF TRANSPORT | 13 | 1804 | 0.0002381 | 0.004118 |
| 270 | NEGATIVE REGULATION OF GROWTH | 5 | 236 | 0.0002483 | 0.004279 |
| 271 | REGULATION OF MITOCHONDRIAL MEMBRANE POTENTIAL | 3 | 54 | 0.0002953 | 0.005051 |
| 272 | NEGATIVE REGULATION OF PEPTIDASE ACTIVITY | 5 | 245 | 0.0002951 | 0.005051 |
| 273 | PROTEIN UBIQUITINATION INVOLVED IN UBIQUITIN DEPENDENT PROTEIN CATABOLIC PROCESS | 4 | 134 | 0.0002983 | 0.005084 |
| 274 | REGULATION OF B CELL PROLIFERATION | 3 | 55 | 0.0003118 | 0.005295 |
| 275 | RESPONSE TO ETHANOL | 4 | 136 | 0.0003156 | 0.00534 |
| 276 | POSITIVE REGULATION OF DEVELOPMENTAL PROCESS | 10 | 1142 | 0.000317 | 0.005344 |
| 277 | CIRCADIAN RHYTHM | 4 | 137 | 0.0003245 | 0.005452 |
| 278 | POSITIVE REGULATION OF TRANSPORT | 9 | 936 | 0.0003348 | 0.005604 |
| 279 | POSITIVE REGULATION OF CATABOLIC PROCESS | 6 | 395 | 0.0003478 | 0.005801 |
| 280 | LEUKOCYTE CELL CELL ADHESION | 5 | 255 | 0.0003546 | 0.005893 |
| 281 | PROTEIN PHOSPHORYLATION | 9 | 944 | 0.0003564 | 0.005902 |
| 282 | DEOXYRIBONUCLEOTIDE BIOSYNTHETIC PROCESS | 2 | 12 | 0.0003666 | 0.006049 |
| 283 | MACROMOLECULAR COMPLEX ASSEMBLY | 11 | 1398 | 0.0003809 | 0.006263 |
| 284 | REGULATION OF EPITHELIAL CELL APOPTOTIC PROCESS | 3 | 59 | 0.0003838 | 0.006284 |
| 285 | CELL ACTIVATION | 7 | 568 | 0.0003849 | 0.006284 |
| 286 | REGULATION OF IMMUNE SYSTEM PROCESS | 11 | 1403 | 0.0003927 | 0.006389 |
| 287 | LEUKOCYTE HOMEOSTASIS | 3 | 60 | 0.0004033 | 0.006538 |
| 288 | MITOTIC CELL CYCLE ARREST | 2 | 13 | 0.0004326 | 0.006917 |
| 289 | REGULATION OF IRE1 MEDIATED UNFOLDED PROTEIN RESPONSE | 2 | 13 | 0.0004326 | 0.006917 |
| 290 | POSITIVE REGULATION OF ENDOPLASMIC RETICULUM UNFOLDED PROTEIN RESPONSE | 2 | 13 | 0.0004326 | 0.006917 |
| 291 | REGULATION OF HISTONE PHOSPHORYLATION | 2 | 13 | 0.0004326 | 0.006917 |
| 292 | RESPONSE TO TEMPERATURE STIMULUS | 4 | 148 | 0.000435 | 0.006932 |
| 293 | LEUKOCYTE ACTIVATION | 6 | 414 | 0.0004463 | 0.007088 |
| 294 | REGULATION OF MULTICELLULAR ORGANISMAL DEVELOPMENT | 12 | 1672 | 0.0004554 | 0.007207 |
| 295 | POSITIVE REGULATION OF CHROMOSOME ORGANIZATION | 4 | 150 | 0.0004576 | 0.007218 |
| 296 | REGULATION OF CELL DIVISION | 5 | 272 | 0.0004761 | 0.007485 |
| 297 | MITOCHONDRIAL DNA METABOLIC PROCESS | 2 | 14 | 0.0005039 | 0.00779 |
| 298 | REGULATION OF FIBRINOLYSIS | 2 | 14 | 0.0005039 | 0.00779 |
| 299 | POSITIVE REGULATION OF EXTRINSIC APOPTOTIC SIGNALING PATHWAY IN ABSENCE OF LIGAND | 2 | 14 | 0.0005039 | 0.00779 |
| 300 | DETERMINATION OF ADULT LIFESPAN | 2 | 14 | 0.0005039 | 0.00779 |
| 301 | REGULATION OF SMOOTH MUSCLE CELL APOPTOTIC PROCESS | 2 | 14 | 0.0005039 | 0.00779 |
| 302 | REGULATION OF RESPONSE TO OXIDATIVE STRESS | 3 | 65 | 0.0005105 | 0.007865 |
| 303 | REGULATION OF CHROMOSOME ORGANIZATION | 5 | 278 | 0.0005258 | 0.008074 |
| 304 | ACTIVATION OF PROTEIN KINASE ACTIVITY | 5 | 279 | 0.0005344 | 0.00818 |
| 305 | REGULATION OF SISTER CHROMATID SEGREGATION | 3 | 67 | 0.000558 | 0.008512 |
| 306 | PHOSPHORYLATION | 10 | 1228 | 0.0005634 | 0.008566 |
| 307 | REGULATION OF JNK CASCADE | 4 | 159 | 0.00057 | 0.008639 |
| 308 | PROTEIN OLIGOMERIZATION | 6 | 434 | 0.0005724 | 0.008647 |
| 309 | DENTATE GYRUS DEVELOPMENT | 2 | 15 | 0.0005805 | 0.008652 |
| 310 | NEGATIVE REGULATION OF B CELL PROLIFERATION | 2 | 15 | 0.0005805 | 0.008652 |
| 311 | RESPONSE TO VITAMIN E | 2 | 15 | 0.0005805 | 0.008652 |
| 312 | NEGATIVE REGULATION OF DEVELOPMENTAL PROCESS | 8 | 801 | 0.000582 | 0.008652 |
| 313 | T CELL APOPTOTIC PROCESS | 2 | 15 | 0.0005805 | 0.008652 |
| 314 | POSITIVE REGULATION OF GENE EXPRESSION | 12 | 1733 | 0.0006296 | 0.009329 |
| 315 | PROTEIN LOCALIZATION TO MITOCHONDRION | 3 | 70 | 0.0006344 | 0.009371 |
| 316 | POSITIVE REGULATION OF CELL PROLIFERATION | 8 | 814 | 0.000647 | 0.009527 |
| 317 | REGULATION OF MAMMARY GLAND EPITHELIAL CELL PROLIFERATION | 2 | 16 | 0.0006625 | 0.009633 |
| 318 | NEGATIVE REGULATION OF ADHERENS JUNCTION ORGANIZATION | 2 | 16 | 0.0006625 | 0.009633 |
| 319 | LEUKOCYTE DIFFERENTIATION | 5 | 292 | 0.0006567 | 0.009633 |
| 320 | REGULATION OF PROTEIN HOMOOLIGOMERIZATION | 2 | 16 | 0.0006625 | 0.009633 |
| Num | GO | Overlap | Size | P Value | Adj. P Value |
|---|---|---|---|---|---|
| 1 | CYCLIN DEPENDENT PROTEIN SERINE THREONINE KINASE REGULATOR ACTIVITY | 6 | 28 | 5.003e-11 | 4.647e-08 |
| 2 | KINASE BINDING | 12 | 606 | 1.381e-08 | 6.416e-06 |
| 3 | ENZYME BINDING | 18 | 1737 | 4.195e-08 | 1.299e-05 |
| 4 | P53 BINDING | 5 | 67 | 5.552e-07 | 0.0001289 |
| 5 | PROTEIN COMPLEX BINDING | 12 | 935 | 1.489e-06 | 0.0002305 |
| 6 | CYCLIN DEPENDENT PROTEIN KINASE ACTIVITY | 4 | 34 | 1.284e-06 | 0.0002305 |
| 7 | KINASE REGULATOR ACTIVITY | 6 | 186 | 5.294e-06 | 0.0007026 |
| 8 | CYCLIN BINDING | 3 | 19 | 1.224e-05 | 0.001421 |
| 9 | PROTEIN KINASE ACTIVITY | 9 | 640 | 1.826e-05 | 0.001697 |
| 10 | MACROMOLECULAR COMPLEX BINDING | 13 | 1399 | 1.735e-05 | 0.001697 |
| 11 | KINASE ACTIVITY | 10 | 842 | 2.531e-05 | 0.002137 |
| 12 | ENZYME REGULATOR ACTIVITY | 10 | 959 | 7.602e-05 | 0.005885 |
| 13 | PROTEIN SERINE THREONINE KINASE ACTIVITY | 7 | 445 | 8.62e-05 | 0.00616 |
| 14 | TRANSFERASE ACTIVITY TRANSFERRING PHOSPHORUS CONTAINING GROUPS | 10 | 992 | 0.0001007 | 0.006683 |
| Num | GO | Overlap | Size | P Value | Adj. P Value |
|---|---|---|---|---|---|
| 1 | CYCLIN DEPENDENT PROTEIN KINASE HOLOENZYME COMPLEX | 7 | 31 | 7.31e-13 | 4.269e-10 |
| 2 | PROTEIN KINASE COMPLEX | 7 | 90 | 1.868e-09 | 5.454e-07 |
| 3 | CATALYTIC COMPLEX | 13 | 1038 | 6.498e-07 | 0.0001265 |
| 4 | TRANSFERASE COMPLEX TRANSFERRING PHOSPHORUS CONTAINING GROUPS | 7 | 237 | 1.462e-06 | 0.0002135 |
| 5 | TRANSFERASE COMPLEX | 9 | 703 | 3.818e-05 | 0.004459 |
Over-represented Pathway
| Num | Pathway | Pathview | Overlap | Size | P Value | Adj. P Value |
|---|---|---|---|---|---|---|
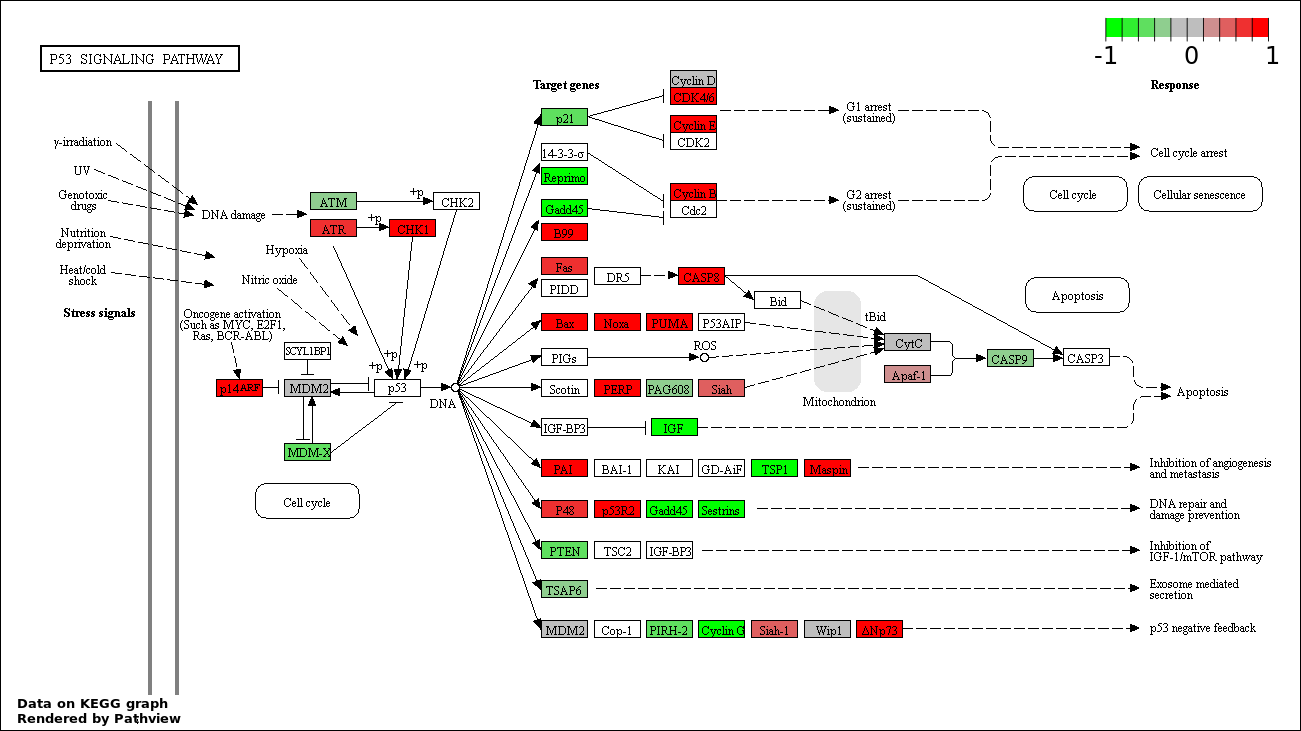
| 1 | hsa04115_p53_signaling_pathway | 48 | 69 | 1.259e-128 | 2.266e-126 | |
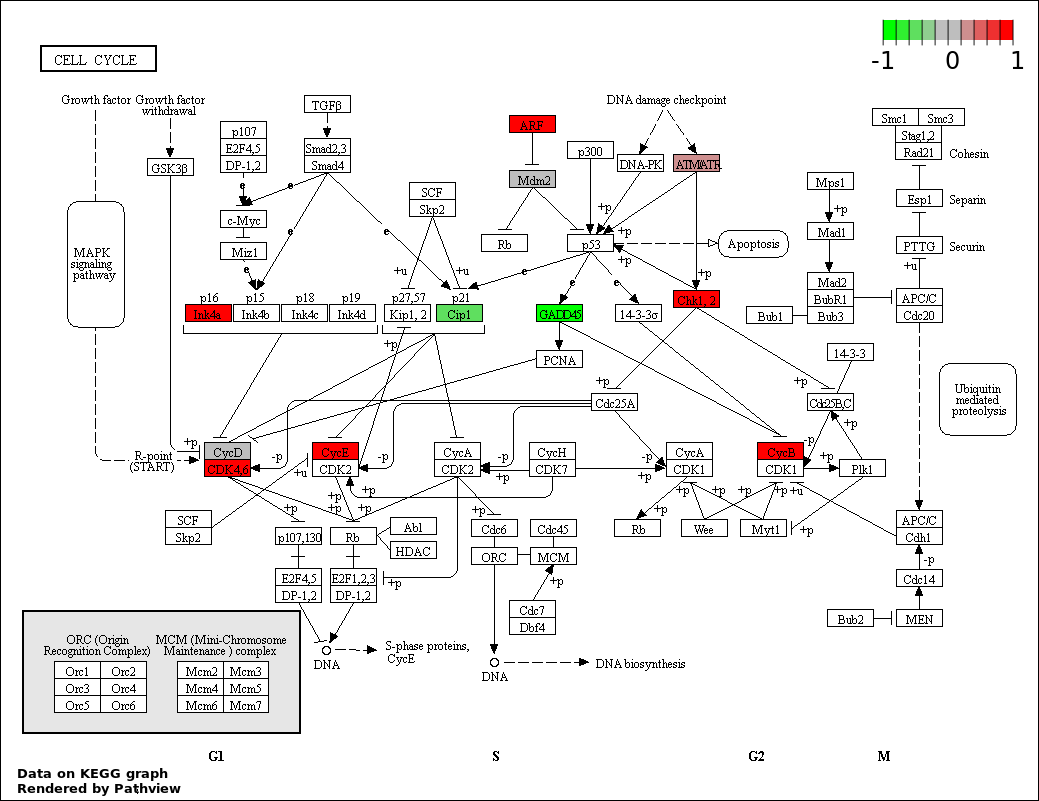
| 2 | hsa04110_Cell_cycle | 17 | 128 | 6.058e-26 | 5.452e-24 | |
| 3 | hsa04151_PI3K_AKT_signaling_pathway | 13 | 351 | 1.332e-12 | 7.993e-11 | |
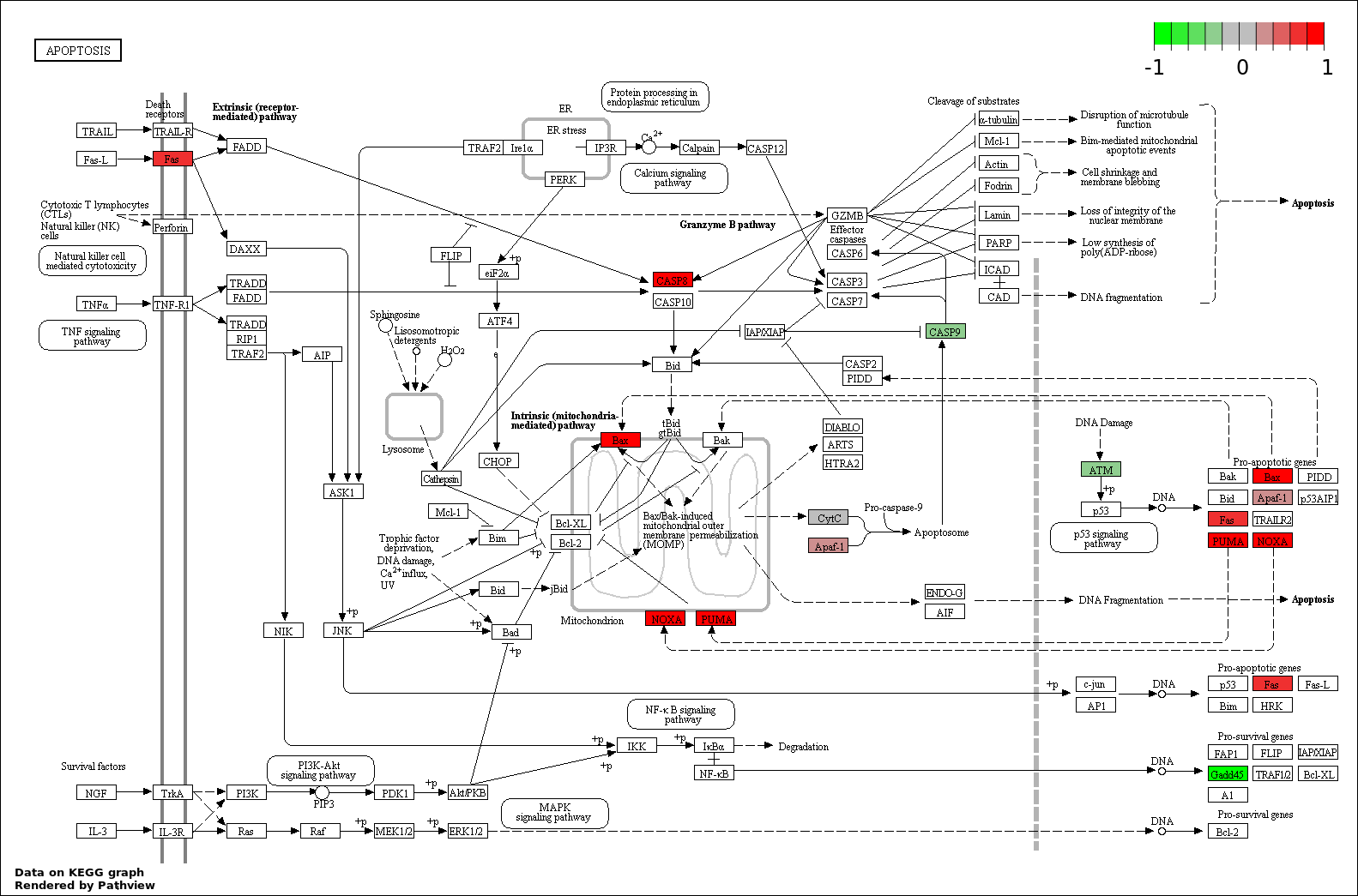
| 4 | hsa04210_Apoptosis | 7 | 89 | 1.726e-09 | 7.766e-08 | |
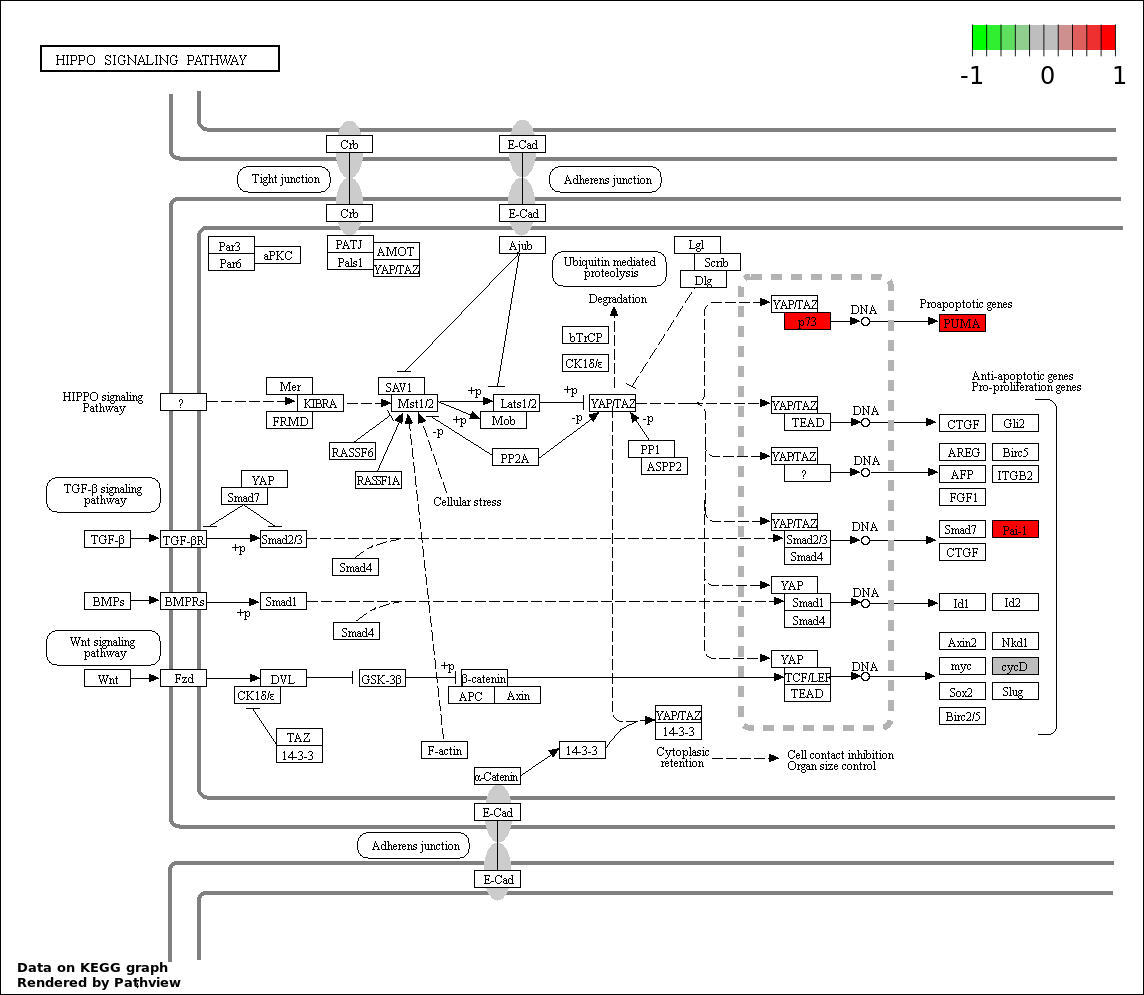
| 5 | hsa04390_Hippo_signaling_pathway | 6 | 154 | 1.776e-06 | 6.394e-05 | |
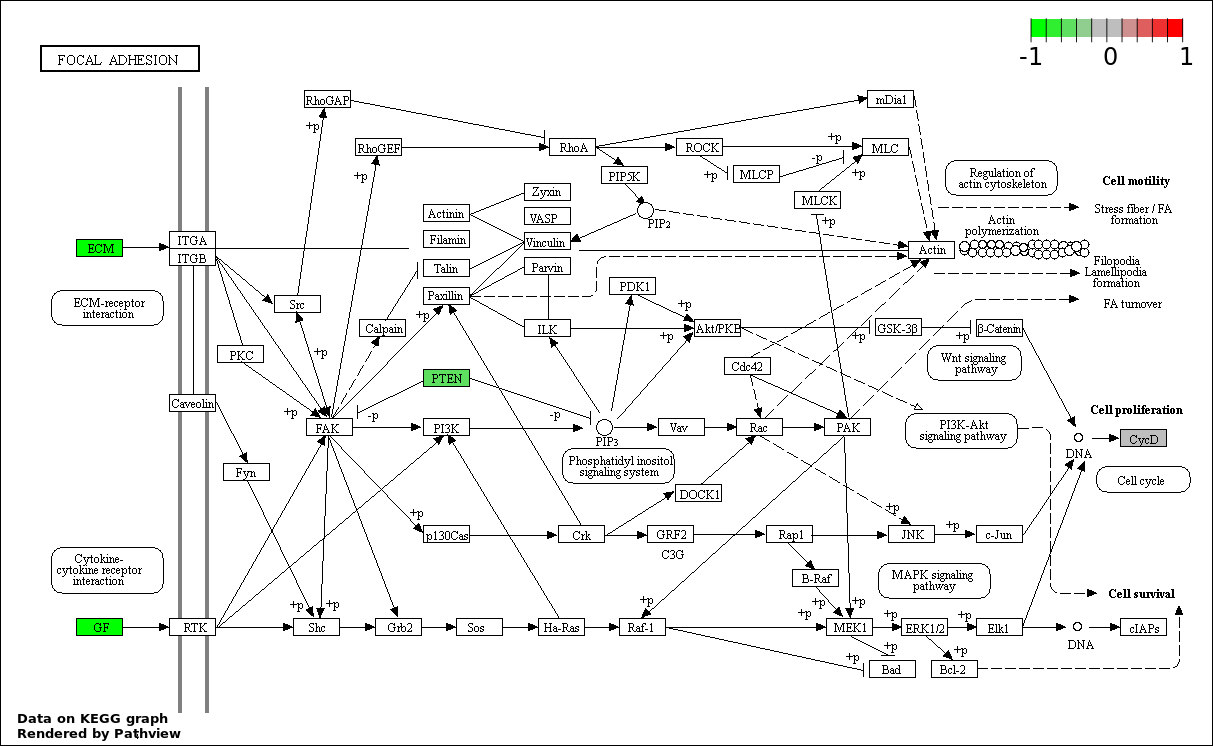
| 6 | hsa04510_Focal_adhesion | 6 | 200 | 8.025e-06 | 0.0002407 | |
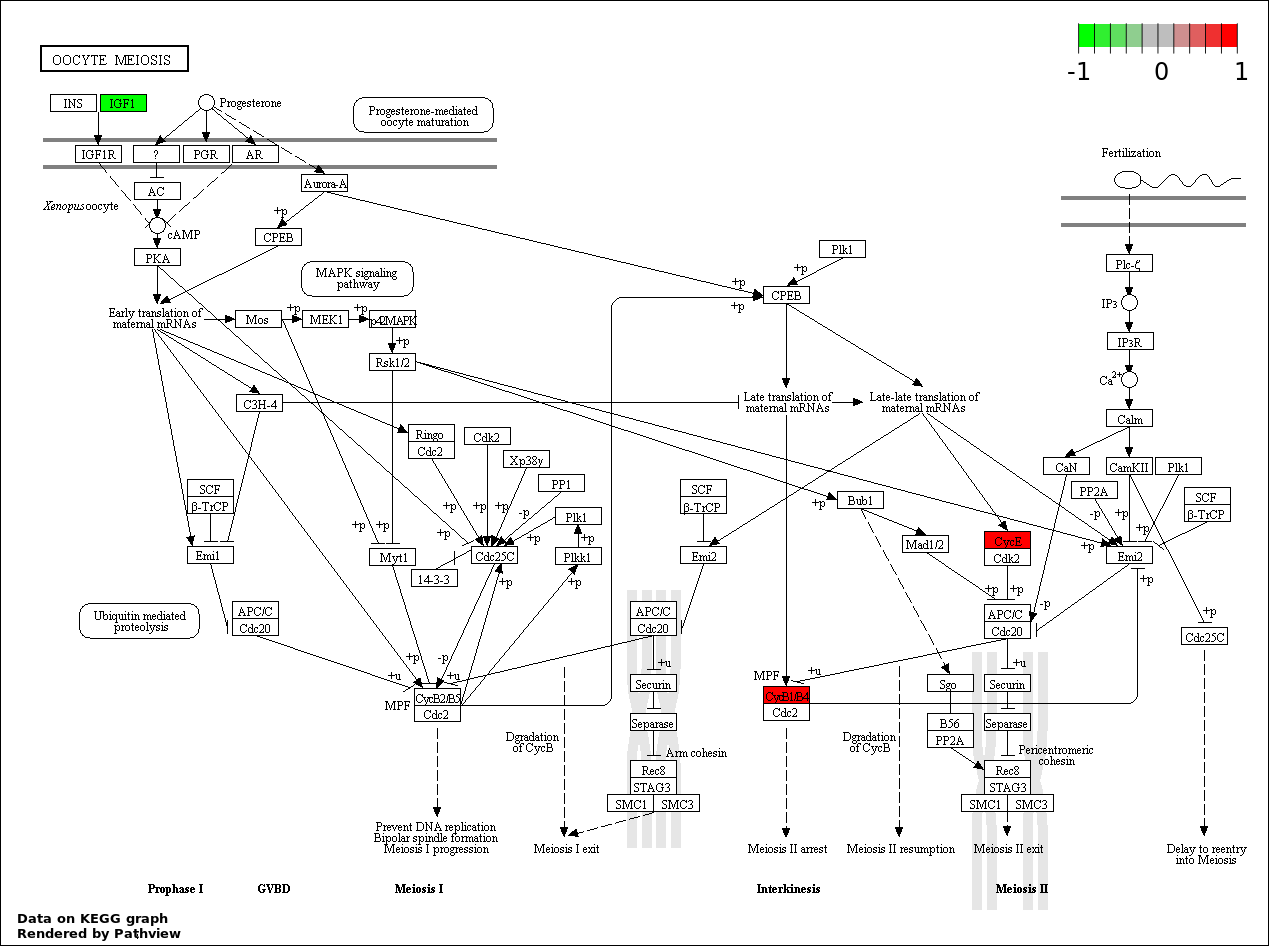
| 7 | hsa04114_Oocyte_meiosis | 4 | 114 | 0.0001606 | 0.004128 | |
| 8 | hsa04120_Ubiquitin_mediated_proteolysis | 4 | 139 | 0.0003429 | 0.007716 | |
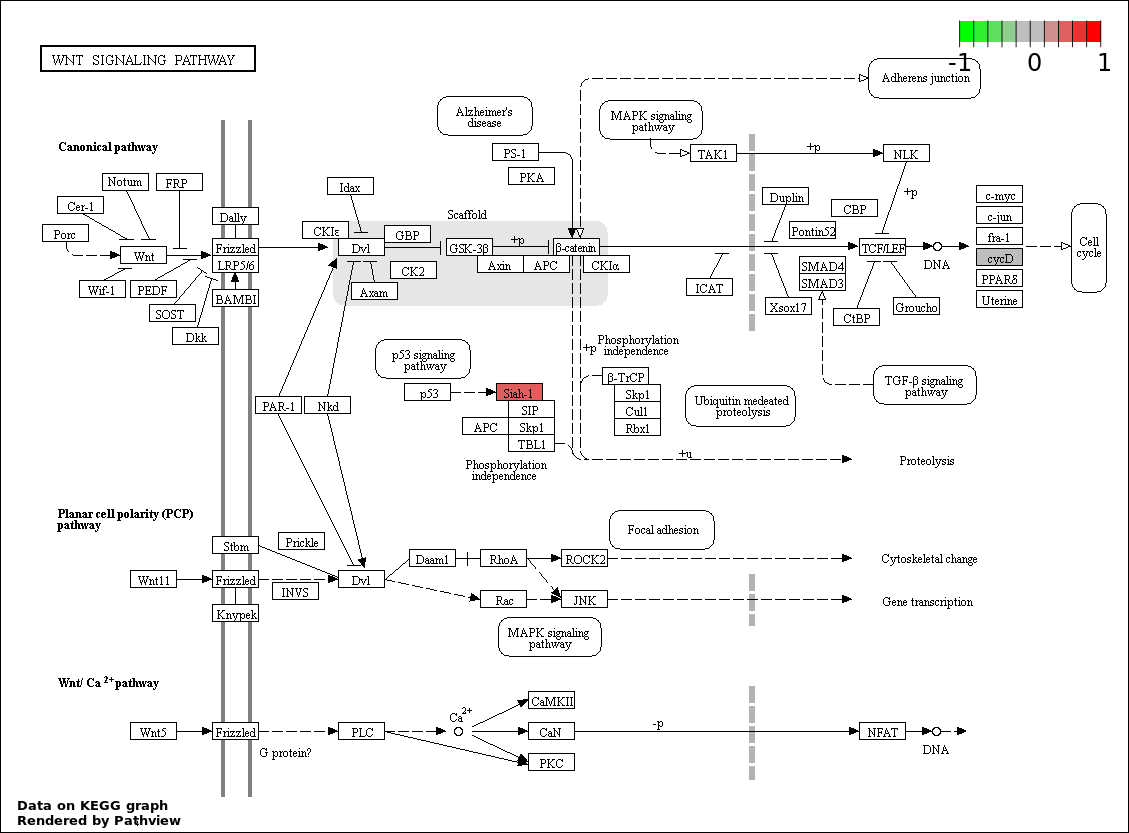
| 9 | hsa04310_Wnt_signaling_pathway | 4 | 151 | 0.0004693 | 0.009385 | |
| 10 | hsa04010_MAPK_signaling_pathway | 4 | 268 | 0.003862 | 0.06952 | |
| 11 | hsa04630_Jak.STAT_signaling_pathway | 3 | 155 | 0.006118 | 0.09631 | |
| 12 | hsa00480_Glutathione_metabolism | 2 | 50 | 0.00642 | 0.09631 | |
| 13 | hsa04914_Progesterone.mediated_oocyte_maturation | 2 | 87 | 0.01854 | 0.2566 | |
| 14 | hsa00240_Pyrimidine_metabolism | 2 | 99 | 0.0236 | 0.3035 | |
| 15 | hsa04722_Neurotrophin_signaling_pathway | 2 | 127 | 0.03732 | 0.4478 | |
| 16 | hsa04530_Tight_junction | 2 | 133 | 0.04058 | 0.4565 | |
| 17 | hsa00230_Purine_metabolism | 2 | 162 | 0.05772 | 0.6111 |
lncRNA-mediated sponge
| Num | lncRNA | miRNAs | miRNAs count | Gene | Sponge regulatory network | lncRNA log2FC | lncRNA pvalue | Gene log2FC | Gene pvalue | lncRNA-gene Pearson correlation |
|---|---|---|---|---|---|---|---|---|---|---|
| 1 | MEG3 |
hsa-let-7d-5p;hsa-miR-151a-3p;hsa-miR-16-1-3p;hsa-miR-17-5p;hsa-miR-182-5p;hsa-miR-18a-5p;hsa-miR-19a-3p;hsa-miR-19b-3p;hsa-miR-200a-5p;hsa-miR-200b-5p;hsa-miR-20a-5p;hsa-miR-335-3p;hsa-miR-33a-3p;hsa-miR-577;hsa-miR-616-5p;hsa-miR-629-3p;hsa-miR-7-1-3p | 17 | THBS1 | Sponge network | -1.645 | 0.00049 | -1.249 | 0.00266 | 0.524 |
| 2 | MEG3 |
hsa-miR-130b-3p;hsa-miR-15b-3p;hsa-miR-16-1-3p;hsa-miR-19a-3p;hsa-miR-19b-1-5p;hsa-miR-19b-3p;hsa-miR-20a-3p;hsa-miR-320b;hsa-miR-33a-3p;hsa-miR-3662;hsa-miR-577;hsa-miR-592;hsa-miR-629-5p | 13 | IGF1 | Sponge network | -1.645 | 0.00049 | -2.083 | 0.00135 | 0.434 |
| 3 | PART1 |
hsa-miR-17-5p;hsa-miR-181b-5p;hsa-miR-186-5p;hsa-miR-192-3p;hsa-miR-19b-1-5p;hsa-miR-20a-5p;hsa-miR-29a-5p;hsa-miR-29b-1-5p;hsa-miR-335-3p;hsa-miR-338-5p;hsa-miR-550a-5p;hsa-miR-577;hsa-miR-625-3p;hsa-miR-625-5p;hsa-miR-7-1-3p;hsa-miR-877-5p | 16 | SESN3 | Sponge network | -2.298 | 0.00168 | 0.054 | 0.92794 | 0.407 |
| 4 | PCA3 |
hsa-let-7a-3p;hsa-let-7b-3p;hsa-let-7f-1-3p;hsa-miR-103a-2-5p;hsa-miR-130b-3p;hsa-miR-15b-3p;hsa-miR-16-1-3p;hsa-miR-181b-5p;hsa-miR-186-5p;hsa-miR-19a-3p;hsa-miR-19b-1-5p;hsa-miR-19b-3p;hsa-miR-20a-3p;hsa-miR-27a-3p;hsa-miR-301a-3p;hsa-miR-32-3p;hsa-miR-320b;hsa-miR-33a-3p;hsa-miR-3662;hsa-miR-454-3p;hsa-miR-576-5p;hsa-miR-590-3p;hsa-miR-629-5p;hsa-miR-940 | 24 | IGF1 | Sponge network | -2.778 | 8.0E-5 | -2.083 | 0.00135 | 0.404 |
| 5 | AGAP11 |
hsa-miR-106b-5p;hsa-miR-17-5p;hsa-miR-181b-5p;hsa-miR-192-3p;hsa-miR-19b-1-5p;hsa-miR-200a-5p;hsa-miR-200b-5p;hsa-miR-20a-5p;hsa-miR-25-3p;hsa-miR-26b-3p;hsa-miR-29a-5p;hsa-miR-29b-1-5p;hsa-miR-320b;hsa-miR-335-3p | 14 | SESN3 | Sponge network | -1.728 | 0.00016 | 0.054 | 0.92794 | 0.346 |
| 6 | AGAP11 |
hsa-let-7f-1-3p;hsa-miR-103a-2-5p;hsa-miR-130b-3p;hsa-miR-15b-3p;hsa-miR-16-1-3p;hsa-miR-181b-5p;hsa-miR-19a-3p;hsa-miR-19b-1-5p;hsa-miR-19b-3p;hsa-miR-20a-3p;hsa-miR-320b;hsa-miR-3662;hsa-miR-421;hsa-miR-629-5p | 14 | IGF1 | Sponge network | -1.728 | 0.00016 | -2.083 | 0.00135 | 0.332 |
| 7 | PCA3 |
hsa-let-7a-3p;hsa-let-7b-3p;hsa-miR-106b-5p;hsa-miR-130b-5p;hsa-miR-141-3p;hsa-miR-15b-5p;hsa-miR-16-2-3p;hsa-miR-16-5p;hsa-miR-181a-2-3p;hsa-miR-182-5p;hsa-miR-183-5p;hsa-miR-19b-3p;hsa-miR-224-3p;hsa-miR-301a-3p;hsa-miR-3065-3p;hsa-miR-424-5p;hsa-miR-590-3p;hsa-miR-590-5p;hsa-miR-93-5p;hsa-miR-96-5p | 20 | CCND2 | Sponge network | -2.778 | 8.0E-5 | -0.496 | 0.3 | 0.318 |
| 8 | PCA3 |
hsa-let-7a-3p;hsa-let-7d-5p;hsa-miR-16-1-3p;hsa-miR-17-5p;hsa-miR-182-5p;hsa-miR-186-5p;hsa-miR-18a-5p;hsa-miR-19a-3p;hsa-miR-19b-3p;hsa-miR-200a-5p;hsa-miR-200b-5p;hsa-miR-20a-5p;hsa-miR-33a-3p;hsa-miR-576-5p;hsa-miR-590-3p;hsa-miR-590-5p;hsa-miR-616-5p;hsa-miR-629-3p;hsa-miR-940 | 19 | THBS1 | Sponge network | -2.778 | 8.0E-5 | -1.249 | 0.00266 | 0.302 |
| 9 | RFPL1S |
hsa-miR-106b-5p;hsa-miR-126-5p;hsa-miR-192-3p;hsa-miR-19b-1-5p;hsa-miR-200a-5p;hsa-miR-200b-3p;hsa-miR-200b-5p;hsa-miR-26b-3p;hsa-miR-29a-5p;hsa-miR-29b-1-5p;hsa-miR-30d-3p;hsa-miR-320b;hsa-miR-335-5p;hsa-miR-429;hsa-miR-550a-5p;hsa-miR-625-5p;hsa-miR-7-1-3p;hsa-miR-877-5p | 18 | SESN3 | Sponge network | -0.223 | 0.70704 | 0.054 | 0.92794 | 0.298 |
| 10 | EMX2OS |
hsa-let-7d-5p;hsa-miR-151a-3p;hsa-miR-17-5p;hsa-miR-182-5p;hsa-miR-18a-5p;hsa-miR-19a-3p;hsa-miR-200a-5p;hsa-miR-200b-5p;hsa-miR-20a-5p;hsa-miR-335-3p;hsa-miR-577;hsa-miR-7-1-3p;hsa-miR-92a-3p | 13 | THBS1 | Sponge network | -1.088 | 0.10042 | -1.249 | 0.00266 | 0.277 |